Summary: A new, non-invasive neuroimaging technique allowed researchers to investigate the visual sensory thalamus, a brain area associated with visual difficulties in dyslexia and other disorders.
Source: TU Dresden
The visual sensory thalamus is a key region that connects the eyes with the cerebral cortex. It contains two major compartments. Symptoms of many diseases are associated with alterations in this region. So far, it has been very difficult to assess these two compartments in living humans, because they are tiny and located very deep inside the brain.
This difficulty of investigating the visual sensory thalamus in detail has hampered the understanding of the function of visual sensory processing tremendously in the past. By coincidence, Christa Müller-Axt, Ph.D. student in the lab of neuroscientist Prof. Katharina von Kriegstein at TU Dresden, discovered structures that she thought might resemble the two visual sensory thalamus compartments in neuroimaging data.
The neuroimaging data was unique because it had an unprecedented high spatial resolution obtained on a specialised magnetic resonance imaging (MRI) machine at the MPI-CBS in Leipzig, where the group was researching developmental dyslexia.
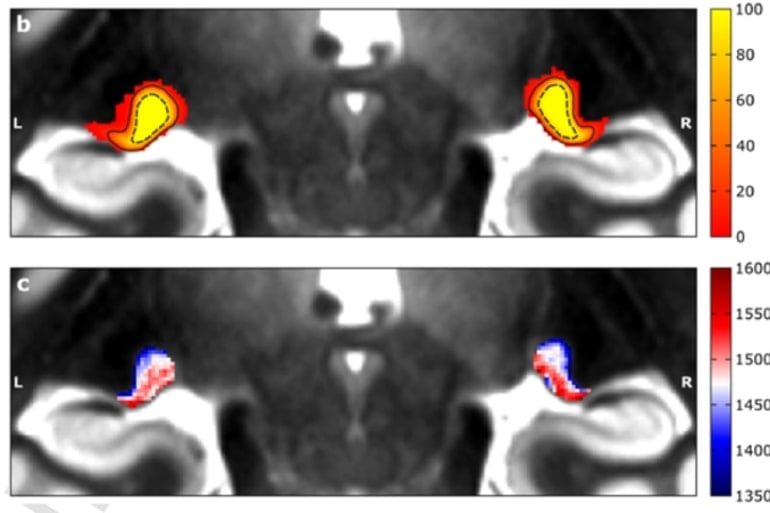
She followed up this discovery in a series of novel experiments involving analysis of high spatial resolution in-vivo and post-mortem MRI data as well as post-mortem histology and was soon sure to have discovered the two major compartments of the visual sensory thalamus.
The results showed that the two major compartments of the visual sensory thalamus are characterised by different amounts of brain white matter (myelin). This information can be detected in novel MRI data and thus, can be used to investigate the two compartments of the visual sensory thalamus in living humans.
“The finding that we can display visual sensory thalamus compartments in living humans is fantastic, as it will be a great tool for understanding visual sensory processing both in health and disease in the near future”, claims first author Christa Müller-Axt and explains,
“Post-mortem studies in developmental dyslexia have shown that there are alterations specifically in one of the two compartments of the visual sensory thalamus. However, there are very few of these post-mortem studies, so it is difficult to say whether all dyslexics are characterised by these kind of visual sensory thalamus alterations.
“Also, post-mortem data cannot reveal anything about the functional impact of these alterations and their specific contribution to developmental dyslexia symptoms.
“Therefore, we expect that our novel in-vivo approach will be a great asset in facilitating research on the role of the visual sensory thalamus in developmental dyslexia.”
About this visual neuroscience research
Author: Anne-Stephanie Vetter
Source: TU Dresden
Contact: Anne-Stephanie Vetter – TU Dresden
Image: The image is credited to TU Dresden
Original Research: Open access.
“Mapping the human lateral geniculate nucleus and its cytoarchitectonic subdivisions using quantitative MRI” by Christa Müller-Axt, Cornelius Eichner, Henriette Rusch, Louise Kauffmann, Pierre-Louis Bazin, Alfred Anwander, Markus Morawski, Katharina von Kriegstein. NeuroImage
Abstract
Mapping the human lateral geniculate nucleus and its cytoarchitectonic subdivisions using quantitative MRI
The human lateral geniculate nucleus (LGN) of the visual thalamus is a key subcortical processing site for visual information analysis. Due to its small size and deep location within the brain, a non-invasive characterization of the LGN and its microstructurally distinct magnocellular (M) and parvocellular (P) subdivisions in humans is challenging.
Here, we investigated whether structural quantitative MRI (qMRI) methods that are sensitive to underlying microstructural tissue features enable MR-based mapping of human LGN M and P subdivisions.
We employed high-resolution 7 Tesla in-vivo qMRI in N = 27 participants and ultra-high resolution 7 Tesla qMRI of a post-mortem human LGN specimen. We found that a quantitative assessment of the LGN and its subdivisions is possible based on microstructure-informed qMRI contrast alone. In both the in-vivo and post-mortem qMRI data, we identified two components of shorter and longer longitudinal relaxation time (T1) within the LGN that coincided with the known anatomical locations of a dorsal P and a ventral M subdivision, respectively.
Through ground-truth histological validation, we further showed that the microstructural MRI contrast within the LGN pertains to cyto- and myeloarchitectonic tissue differences between its subdivisions. These differences were based on cell and myelin density, but not on iron content.
Our qMRI-based mapping strategy paves the way for an in-depth understanding of LGN function and microstructure in humans. It further enables investigations into the selective contributions of LGN subdivisions to human behavior in health and disease.






