Summary: Mapping the functional units of the vinegar fly’s brain, researchers discover the olfactory glomeruli differs in structure.
Source: Max Planck Institute.
The basic units of the olfactory system in the fly brain provide references to their function and ecological relevance.
Scientists at the Max Planck Institute for Chemical Ecology in Jena, Germany, have now quantified and mapped the functional units of the olfactory center in the brains of vinegar flies responsible for the perception of odors. They found out that the so-called olfactory glomeruli in the antennal lobe, the insect analogue of the olfactory bulb of mammals, differ from each other in their architecture. The morphology and the structure of these spherical brain units provide information about the ecological relevance of the odors they process, especially with regard to the flies’ odor-guided behavior.
When insects search for food, a sexual mate or the ideal place for laying eggs – somewhere where the hatching larvae have a good chance to survive – they have to rely on their sense of smell. They use their olfactory organs, the antennae, to detect odor molecules in their environment. These odors are processed in the so-called antennal lobe, the actual olfactory center of the insect brain, which consists of spherical structures: the olfactory glomeruli. Here, inside the olfactory glomeruli, different groups of olfactory neurons form conjunctions or synapses which enable different environmental odors to be processed.
Until now, scientists had assumed that olfactory glomeruli share a pretty homogeneous architecture and that particular functions of the different glomeruli can be attributed primarily to special olfactory receptors on the membranes of the olfactory sensory neurons. A research team of the Department of Evolutionary Neuroethology has now shown for the first time that the neuronal composition of each glomerulus is unique and highly specific. “Each glomerulus processes different odors in a unique way. The odors, on the other hand, differ in their impact on the behavior of the vinegar flies,” explains Silke Sachse, the leader of the study
The researchers scrutinized the neuronal architecture of the olfactory glomeruli in the vinegar fly Drosophila melanogaster and linked the morphological differences they observed to the respective functions. They labeled different neurons in single glomeruli, and counted and anatomically described these. In order to achieve this, they used transgenic Drosophila lines which expressed the photoactivatable green fluorescent protein (PA-GFP) in the neurons and high-resolution confocal microscopy.
“As assumed, but until now not experimentally proven, glomeruli which contained many olfactory sensory neurons were generally bigger than were the glomeruli with fewer olfactory sensory neurons. This means that the anatomical shape of a glomerulus already provides information about how sensitive it will respond to a certain odor,” Veit Grabe, one of the first authors of the study, reports. The scientists also investigated whether there were any differences between male and female flies: Glomeruli which processed the Drosophila sex pheromones were bigger in males which were able to perceive the female attractants from large distances. Moreover, the researchers counted the so-called projection neurons which are responsible for the transmission of odor stimuli to higher regions of the olfactory system. Glomeruli containing highly specific olfactory sensory neurons – namely, those which process only one single odor – seem to exhibit more projection neurons than glomeruli which process many different odors. More projection neurons make sure that information is transferred to higher brain regions in a faster and more reliable way to result in rapid odor-guided decisions.
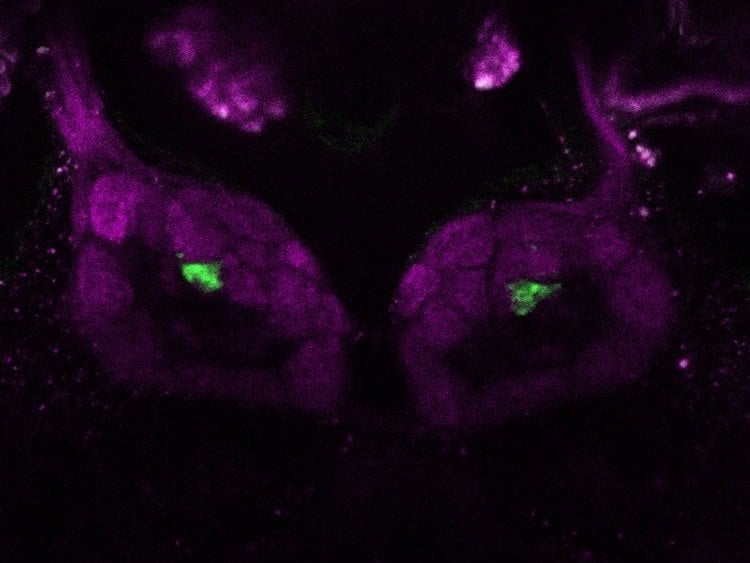
In order to link the morphology of a glomerulus to its function, the research team determined another important parameter by means of electrophysiological analysis: “Lifetime sparseness” indicates how many different odors can activate an olfactory receptor. If a receptor is activated by a large variety of odors, it has very low “sparseness” and is therefore a very broad-range receptor. The higher the “sparseness” of a receptor, the fewer odors it is able to perceive. An extreme case would be a highly specific receptor which responds only to a single odor. Odors which are very attractive to vinegar flies include their own pheromones, but also odors which provide clues about where to find a food source, such as overripe fruit. Some of these attractive odors may elicit oviposition in females, because they provide information about a place where larvae can hatch and have a good chance to survive. However, important odors may also be deterrents – and warning signals – because they contain information about lethal dangers, such as toxic substances in a potential food source or parasites.
“The complete quantitative mapping of all olfactory sensory neurons linked together in each olfactory glomerulus helped us to create an extended morphological basis for a better understanding of the role and function these units in the olfactory system have,” co-author Amelie Baschwitz summarizes.
Because these new insights are not limited to the vinegar fly, and may also apply to other animals or even humans, they have far-reaching significance.
Source: Silke Sachse – Max Planck Institute
Image Source: NeuroscienceNews.com image is credited to MPI f. Chemical Ecology/ V. Grabe.
Original Research: Full open access research for “Elucidating the neuronal architecture of olfactory glomeruli in the Drosophila antennal lobe” by Grabe, V., Baschwitz, A., Dweck, H. K. M., Lavista-Llanos, S., Hansson, B., and Sachse, S in Cell Reports. Published online September 20 2016 doi:10.1016/j.celrep.2016.08.063
[cbtabs][cbtab title=”MLA”]Max Planck Institute. “Olfactory Glomeruli Have a Unique Structure.” NeuroscienceNews. NeuroscienceNews, 21 September 2016.
<https://neurosciencenews.com/structure-olfactory-glomeruli-5094/>.[/cbtab][cbtab title=”APA”]Max Planck Institute. (2016, September 21). Olfactory Glomeruli Have a Unique Structure. NeuroscienceNews. Retrieved September 21, 2016 from https://neurosciencenews.com/structure-olfactory-glomeruli-5094/[/cbtab][cbtab title=”Chicago”]Max Planck Institute. “Olfactory Glomeruli Have a Unique Structure.” https://neurosciencenews.com/structure-olfactory-glomeruli-5094/ (accessed September 21, 2016).[/cbtab][/cbtabs]
Abstract
Elucidating the neuronal architecture of olfactory glomeruli in the Drosophila antennal lobe
Highlights
•The neuronal composition of glomeruli is unique
•Glomerular volume is determined largely by the number of olfactory sensory neurons
•Selectivity of a glomerulus is represented by the number of second-order neurons
•Glomeruli encoding crucial odors possess a large number of PNs and low LN innervation
Summary
Olfactory glomeruli are morphologically conserved spherical compartments of the olfactory system, distinguishable solely by their chemosensory repertoire, anatomical position, and volume. Little is known, however, about their numerical neuronal composition. We therefore characterized their neuronal architecture and correlated these anatomical features with their functional properties in Drosophila melanogaster. We quantitatively mapped all olfactory sensory neurons (OSNs) innervating each glomerulus, including sexually dimorphic distributions. Our data reveal the impact of OSN number on glomerular dimensions and demonstrate yet unknown sex-specific differences in several glomeruli. Moreover, we quantified uniglomerular projection neurons for each glomerulus, which unraveled a glomerulus-specific numerical innervation. Correlation between morphological features and functional specificity showed that glomeruli innervated by narrowly tuned OSNs seem to possess a larger number of projection neurons and are involved in less lateral processing than glomeruli targeted by broadly tuned OSNs. Our study demonstrates that the neuronal architecture of each glomerulus encoding crucial odors is unique.
“Elucidating the neuronal architecture of olfactory glomeruli in the Drosophila antennal lobe” by Grabe, V., Baschwitz, A., Dweck, H. K. M., Lavista-Llanos, S., Hansson, B., and Sachse, S in Cell Reports. Published online September 20 2016 doi:10.1016/j.celrep.2016.08.063