Summary: A new study challenges 75 year old dogma of mammalian vision. Researchers have shows the post rhinal cortex obtains visual data from the superior colliculus and is not dependent upon information from primary visual cortex.
Source: UCSF.
The visual system is probably the best understood part of the brain. Over the past 75 years, neuroscientists have assembled a detailed account of how light waves entering your eyes allow you to recognize your grandmother’s face, to track a hawk in flight, or to read this sentence. But a new study by UC San Francisco researchers is calling a fundamental aspect of vision science into question, showing that even the best-studied parts of the brain can still hold plenty of surprises.
According to the standard model of visual processing, all visual information from the retina must first pass through the primary visual cortex (V1) in the back of the brain, which extracts simple features like lines and edges, before being distributed to a number of “higher order” visual areas that extract increasingly complex features like shapes, shading, movement, and so on.
The new study — published online January 4, 2019 in Science — shows for the first time that one of these supposedly higher-order visual areas, which is involved in the perception of moving objects, does not depend on information from V1 at all. Instead, this region, known as the post-rhinal cortex (POR), appears to obtain visual data directly from an evolutionarily ancient sensory processing center at the base of the brain called the superior colliculus.
“It’s as if we’ve discovered a second primary visual cortex,” said study senior author Massimo Scanziani, PhD, a professor of physiology at UCSF and a Howard Hughes Medical Institute investigator. “This undermines the whole concept of the visual system in mammalian cortex as a perfect hierarchy with V1 as the gatekeeper and raises a multitude of questions, including how these two parallel visual systems evolved and how they cooperate to produce a unified visual experience.”
What the Lizard Brain Shows the Mammalian Cortex
The ancestral superior colliculus (called optic tectum in non-mammals) is the main sensory processing center in creatures with little or no cortex, such as fish, amphibians, lizards, and birds. It is particularly attuned to motion and tied in to reflexive behaviors — think of a frog’s ability to snag a fly from the air with its tongue or a fish’s ability to dart away from a looming predator.
However, the superior colliculus did not disappear with the development of the cortex in mammals. In primates, including humans, it has been linked to rapid and unconscious forms of visual processing, such as jumping in fright when you see a stick that looks like a snake, or automatically catching a ball thrown at your face. It may also play a role in directing the spotlight of visual attention (such as when you are waiting for a traffic light to turn green or searching a crowded scene for someone in a particular red and white striped hat). But the new paper is the first time anyone has shown that this evolutionarily ancient system has a dedicated space in cortex.
The research was launched when lead author Riccardo Beltramo, PhD, a postdoctoral researcher in the Scanziani lab, was recording neural responses to moving visual stimuli in the mouse POR, which is known to play roles in perceiving motion and in spatial memory. He used a technique called optogenetics to temporarily silence activity in V1 with light, hoping to confirm his expectation that POR responses depend on information flow through the standard visual hierarchy. But to his surprise, he found that POR neurons continued to respond to moving stimuli even without input from V1.
“It was absolutely remarkable,” Beltramo said. “We silenced the main visual area in the cortex and visual responses in POR remained unaffected. That was the first big ‘wow’ moment that told us we were on to something completely unexpected.”
If POR’s responses to moving objects were not coming from V1, Beltramo wondered, then how did they get there? There must be another pathway connecting POR to visual information coming in from the retina, he reasoned. To identify this parallel visual pathway, Beltramo used injections of custom-engineered viruses that label neurons that are connected to one another. This let him show that POR neurons get two sources of anatomical input — one from V1, and a second from the superior colliculus, each routed through distinct zones of the thalamus, the brain’s central relay station.
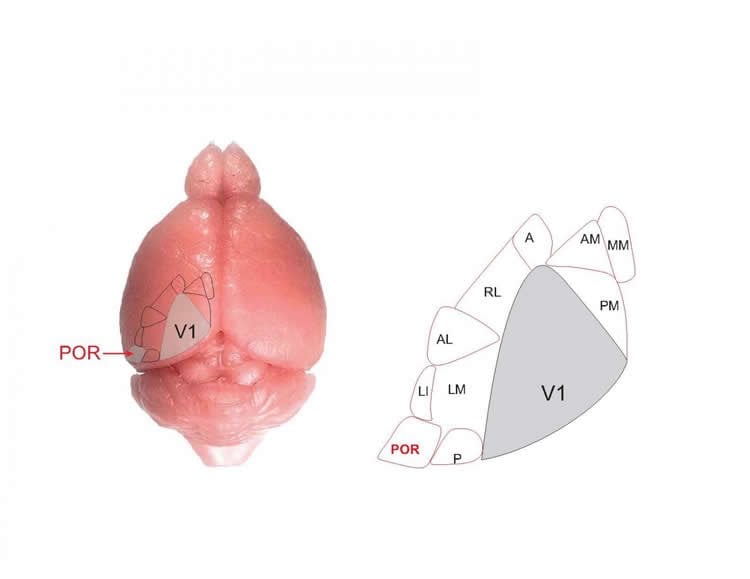
To confirm that the superior colliculus was driving POR responses to movement, Beltramo used optogenetics to systematically silence either V1 or the superior colliculus while recording from POR. He showed that — unlike inactivating V1 — shutting down the superior colliculus made POR activity totally disappear. Indeed, the superior colliculus appears to be critical for POR’s ability to track moving objects, something the researchers found POR’s V1 inputs were unable to do on their own.
Study Raises Fundamental Questions About Brain Evolution, Function
More research is needed to test whether visual responses in POR-like areas of the brains of primates like ourselves also depend on input from the superior colliculs, as well as how V1-driven and colliculus-driven activity interact to influence animals’ behavior, the authors say.
“We hypothesize that POR, which was previously considered a higher order visual area, could be a sort of ‘primal’ visual cortex, similar to a simple early amphibian, reptilian or avian visual cortex, and that it may be dedicated to the detection that something is moving in the environment, whether it is small, close prey or a large distant predator,” Beltramo said. “From this perspective, perhaps V1 would add to this information a more precise discrimination of the nature of the moving object, such as its exact location or whether it is a potentially tasty beetle or a potentially deadly scorpion.”

Based on previous studies, the newly discovered superior colliculus-POR system could also be linked to fear responses, spatial attention and navigation, or even face recognition — all specialties of the region of temporal cortex where POR is located.
The findings could also have implications for an intriguing phenomenon called “blindsight,” in which people who become blind because of damage to V1 are still able to identify the positions of objects and navigate obstacles, even though they cannot consciously perceive them. Based on studies in primates, blindsight is thought depend on the superior colliculus, but the new study suggests it could involve POR-like areas of the cortex as well.
“This is one of those findings that raises a lot of questions rather than answering any, because, as many discoveries, it presents questions no one knew to ask,” Scanziani said.
Funding: The research was supported by the European Molecular Biology Organization Long Term Fellowship, the Human Frontiers Science Program Long Term Fellowship, and the Howard Hughes Medical Institute.
Study senior author Massimo Scanziani, PhD, is a professor of physiology at UCSF. He is also a Howard Hughes Medical Institute Investigator.
Study lead author Riccardo Beltramo, PhD, is a postdoctoral researcher in the Scanziani lab.
Source: Nicholas Weiler – UCSF
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com images are credited to Scanziani lab / UCSF.
Original Research: Open access research for “A collicular visual cortex: Neocortical space for an ancient midbrain visual structure” by Riccardo Beltramo, and Massimo Scanziani in Science. Published January 3 2019.
doi:10.1126/science.aau7052
[cbtabs][cbtab title=”MLA”]UCSF”Surprise Discovery Reveals Second Visual System in Cerebral Cortex: Mouse Study.” NeuroscienceNews. NeuroscienceNews, 3 January 2019.
<https://neurosciencenews.com/second-visual-system-10419/>.[/cbtab][cbtab title=”APA”]UCSF(2019, January 3). Surprise Discovery Reveals Second Visual System in Cerebral Cortex: Mouse Study. NeuroscienceNews. Retrieved January 3, 2019 from https://neurosciencenews.com/second-visual-system-10419/[/cbtab][cbtab title=”Chicago”]UCSF”Surprise Discovery Reveals Second Visual System in Cerebral Cortex: Mouse Study.” https://neurosciencenews.com/second-visual-system-10419/ (accessed January 3, 2019).[/cbtab][/cbtabs]
Abstract
A collicular visual cortex: Neocortical space for an ancient midbrain visual structure
Visual responses in the cerebral cortex are believed to rely on the geniculate input to the primary visual cortex (V1). Indeed, V1 lesions substantially reduce visual responses throughout the cortex. Visual information enters the cortex also through the superior colliculus (SC), but the function of this input on visual responses in the cortex is less clear. SC lesions affect cortical visual responses less than V1 lesions, and no visual cortical area appears to entirely rely on SC inputs. We show that visual responses in a mouse lateral visual cortical area called the postrhinal cortex are independent of V1 and are abolished upon silencing of the SC. This area outperforms V1 in discriminating moving objects. We thus identify a collicular primary visual cortex that is independent of the geniculo-cortical pathway and is capable of motion discrimination.






