Summary: Personalized rTMS treatments could be revolutionary in helping combat neuropsychiatric disorders, researchers say.
Source: SfN.
A study of epilepsy patients with implanted electrodes provides an unprecedented view of the changes in brain activity created by electrical stimulation. These findings, published in Journal of Neuroscience, have the potential to improve noninvasive stimulation approaches toward the treatment of neuropsychiatric disorders.
Repetitive transcranial magnetic stimulation (rTMS) is increasingly used in patients with disorders such as depression that do not respond well to medication or psychotherapy.
Although the effects of stimulation on the motor cortex have been characterized in animal models and humans, its effects on other brain areas — including the prefrontal cortex, the target of rTMS in depression — are unclear.
Corey Keller and colleagues mimicked rTMS of the prefrontal cortex in four epileptic patients who were previously implanted with brain electrodes to manage their condition. This allowed the researchers to study changes in the neural activity of specific regions with a resolution that is not possible with noninvasive brain stimulation and imaging.
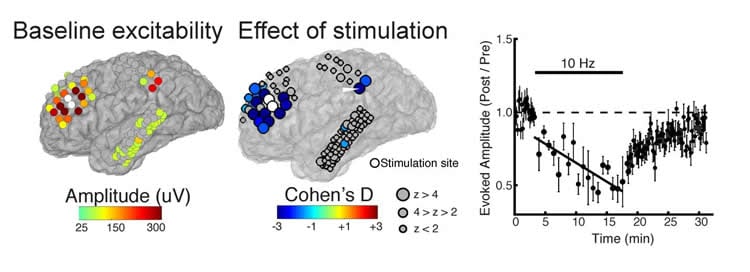
Comparing participants’ brain excitability before and after the rTMS treatment, the team found that they were able to accurately predict which brain regions would be affected by the stimulation. This research could inform the development of individualized stimulation protocols.
Funding: The study was financed by the NIH/National Institute of Neurological Disorders and Stroke.
Source: David Barnstone – SfN
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Corey Keller.
Original Research: Abstract for “Induction and quantification of excitability changes in human cortical networks” by Corey J. Keller, Yuhao Huang, Jose L. Herrero, Maria Fini, Victor Du, Fred A. Lado, Christopher J. Honey and Ashesh D. Mehta in Journal of Neuroscience. Published May 21 2018.
doi:10.1523/JNEUROSCI.1088-17.2018
[cbtabs][cbtab title=”MLA”]SfN “Personalizing Therapeutic Brain Stimulation.” NeuroscienceNews. NeuroscienceNews, 21 May 2018.
<https://neurosciencenews.com/personalized-brain-stimulation-9096/>.[/cbtab][cbtab title=”APA”]SfN (2018, May 21). Personalizing Therapeutic Brain Stimulation. NeuroscienceNews. Retrieved May 21, 2018 from https://neurosciencenews.com/personalized-brain-stimulation-9096/[/cbtab][cbtab title=”Chicago”]SfN “Personalizing Therapeutic Brain Stimulation.” https://neurosciencenews.com/personalized-brain-stimulation-9096/ (accessed May 21, 2018).[/cbtab][/cbtabs]
Abstract
Induction and quantification of excitability changes in human cortical networks
How does human brain stimulation result in lasting changes in cortical excitability? Uncertainty on this question hinders the development of personalized brain stimulation therapies. To characterize how cortical excitability is altered by stimulation, we applied repetitive direct electrical stimulation in eight human subjects (male and female) undergoing intracranial monitoring. We evaluated single-pulse corticocortical evoked potentials (CCEPs) before and after repetitive stimulation across prefrontal (N=4), temporal (N=1), and motor (N=3) cortices. We asked if a single session of repetitive stimulation was sufficient to induce excitability changes across distributed cortical sites. We found a subset of regions at which 10Hz prefrontal repetitive stimulation resulted in both potentiation and suppression of excitability that persisted for at least 10 minutes. We then asked if these dynamics could be modeled by the pre-stimulation connectivity profile of each subject. We found that cortical regions (i) anatomically close to the stimulated site and (ii) exhibiting high-amplitude CCEPs underwent changes in excitability following repetitive stimulation. We demonstrate high accuracy (72-95%) and discriminability (81-99%) in predicting regions exhibiting changes using individual subjects’ pre-stimulation connectivity profile, and show that adding pre-stimulation connectivity features significantly improved model performance. The same features predicted regions of modulation following motor and temporal cortices stimulation in an independent dataset. Taken together, baseline connectivity profile can be used to predict regions susceptible to brain changes and provides a basis for personalizing therapeutic stimulation.
Significance Statement. Brain stimulation is increasingly used to treat neuropsychiatric disorders by inducing excitability changes at specific brain regions. However, our understanding of how, when, and where these changes are induced is critically lacking. We inferred plasticity in the human brain after applying electrical stimulation to the brain’s surface and measuring changes in excitability. We observed excitability changes in regions anatomically and functionally closer to the stimulation site. Those in responsive regions were accurately predicted using a classifier trained on baseline brain network characteristics. Finally, we showed that the excitability changes can potentially be monitored in real-time. These results begin to fill basic gaps in our understanding of stimulation-induced neuronal dynamics in humans and offer pathways to optimize stimulation protocols.