Summary: Losing the ability to read, recognize faces, or see color is the devastating reality for those with age-related macular degeneration (AMD) or inherited retinal diseases. Until now, there has been no approved therapy to stop the death of cone photoreceptors—the cells responsible for high-resolution central vision.
A breakthrough study
has identified specific genetic pathways and molecules that can shield these vital cells from degeneration. The team used a massive “human-in-a-dish” system—testing over 2,700 compounds on 20,000 human retinal organoids—to find a “key” to cone survival.
Key Facts
- The Casein Kinase 1 Breakthrough: Researchers identified that inhibiting casein kinase 1 (CK1) acts as a powerful protective mechanism, keeping cones alive even under disease-mimicking stress.
- Massive Screening: The study is one of the largest of its kind, screening 2,700 compounds to see which helped cones thrive and which caused “retinal toxicity” (damage).
- Cross-Species Validation: The protective molecules weren’t just effective in lab-grown human organoids; they were also confirmed to stop retinal degeneration in living mouse models.
- Public Data Resource: The team has released their entire dataset to the public, providing a “roadmap” for other scientists to develop central vision therapies and avoid toxic drug combinations.
Source: IOB Basel
Scientists led by Botond Roska at the Institute of Molecular and Clinical Ophthalmology Basel (IOB) and collaborators have identified genetic pathways and compounds capable of protecting cone photoreceptors from the degeneration that underlies conditions like age-related macular degeneration.
Cone photoreceptors, concentrated in the macula, are essential for reading, recognizing faces, and perceiving colours. Their death, as it happens in many inherited retinal diseases and macular degeneration, leads to the loss of central vision. Despite decades of research, no approved therapies can halt this process.
This new study, conducted by first authors Stefan Spirig, Alvaro Herrero Navarro and collaborators, addresses this unmet need using a human-based experimental system.
Key findings
- More than 2,700 compounds were tested in 20,000 human retinal organoids
- Some compound classes were found to damage cones, highlighting safety risks
- Several molecules were identified that protect cone photoreceptors from degeneration
- Inhibition of casein kinase 1 emerged as a key protective mechanism
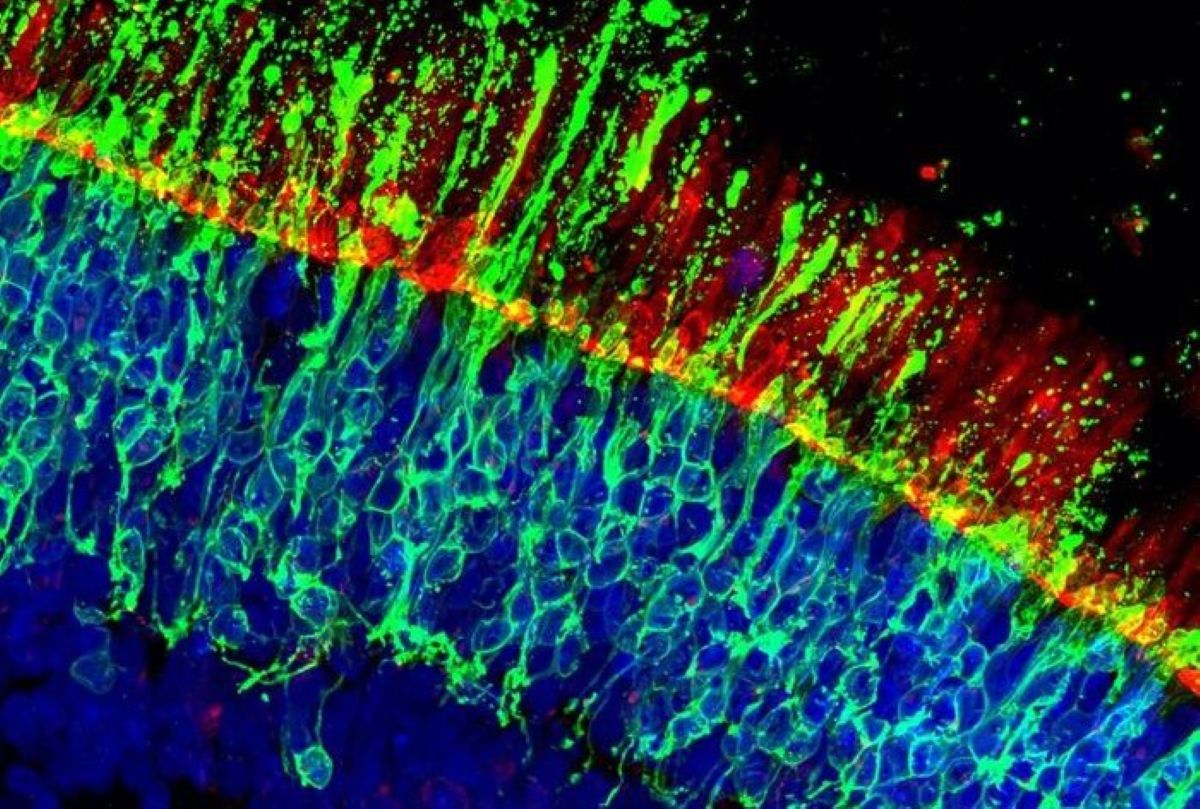
Cone photoreceptors were selectively labelled, allowing their fate to be followed over time under controlled stress conditions that mimic disease. This approach enabled a systematic screen of compounds with known molecular targets.
Clear patterns emerged: two kinase inhibitors consistently protected cones over extended periods. The protective effects held across different stress conditions and were further confirmed in a mouse model of retinal degeneration, supporting their broader relevance.
Beyond identifying protective pathways, the study makes a comprehensive dataset publicly available, covering the compounds tested, their molecular targets, and their effects on human cone survival.
This resource will guide the development of therapies aimed at preserving central vision and enable a systematic assessment of potential retinal toxicity.
By combining retinal biology, organoid technology, and large-scale compound screening, the work gives researchers a head start in developing new treatments and sharpens focus on a long-standing goal in ophthalmology: protecting the very cells that make sight possible.
Key Questions Answered:
A: Cones are high-energy cells concentrated in the macula. Because they are so specialized for color and detail, they are incredibly sensitive to metabolic stress. This study is revolutionary because it used human retinal organoids—miniature lab-grown retinas—to study these cells in a human context rather than just relying on animals.
A: It’s a massive step toward prevention. While it doesn’t “grow back” lost cones, the discovery of CK1 inhibitors provides a way to shield existing cones from dying. For a patient with early-stage AMD, this could mean the difference between keeping their central vision and losing the ability to read or drive.
A: Think of it as a “retina-in-a-dish.” Scientists use stem cells to grow three-dimensional, functional human retinal tissue. This allows them to test thousands of drugs safely and see exactly how human cones react to stress and treatment before ever moving to a human trial.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this visual neuroscience and AMD research news
Author: Elsa Sigle
Source: IOB
Contact: Elsa Sigle – IOB
Image: The image is credited to IOB
Original Research: Open access.
“Cell type-focused compound screen in human organoids reveals CK1 inhibition protects cone photoreceptors from death” by Stefan E. Spirig, Álvaro Herrero-Navarro, Larissa Utz, Valeria J. Arteaga-Moreta, Zoltan Raics, Susana Posada-Céspedes, Stephanie Chreng, Olaf Galuba, Inga Galuba, Isabelle Claerr, Steffen Renner, Miklos Boldogkoi, Verónica Moreno-Juan, P. Timo Kleindienst, Adrienn Volak, Jannick Imbach, Svitlana Malysheva, Rebecca A. Siwicki, Vincent Hahaut, Yanyan Hou, Tiago M. Rodrigues, Simone Picelli, Marco Cattaneo, Josephine Jüttner, Cameron S. Cowan, Myriam Duckely, Daniel K. Baeschlin, Magdalena Renner, Vincent Unterreiner, and Botond Roska. Neuron
DOI:10.1016/j.neuron.2026.02.024
Abstract
Cell type-focused compound screen in human organoids reveals CK1 inhibition protects cone photoreceptors from death
Human organoids that mirror their corresponding organs in cell-type diversity present an opportunity to perform large-scale screens for compounds that protect disease-affected or damaged healthy cell types.
Here, we generated 20,000 human retinal organoids with green fluorescent protein (GFP)-labeled cone photoreceptors. Since degeneration of cones is a leading cause of blindness, we induced cone death and screened 2,707 compounds with known targets for those that saved cones or those that further damaged cones.
We identified inhibitors of casein kinase 1 (CK1) that protected cones, heat shock protein 90 (HSP90) inhibitors that saved cones in the short term but damaged them in the longer term, and broad histone deacetylase (HDAC) inhibition by many compounds that significantly damaged cones.
Finally, we confirmed the protective effects of identified compounds in a mouse model of photoreceptor degeneration.
This work provides a database for cone-damaging compounds and describes compounds and targets that can be starting points to develop neuroprotection for cones in diseases such as macular degeneration.