Summary: A new study reveals head impact, not concussion, triggers CTE. The discovery could help with early detection and treatments for the neurodegenerative disorder.
Source: BMC.
Researchers have identified evidence of early Chronic Traumatic Encephalopathy (CTE) brain pathology after head impact–even in the absence of signs of concussion. Early indicators of CTE pathology not only persisted long after injury but also spread through the brain, providing the best evidence to date that head impact, not concussion, causes CTE.
The findings, published online in the journal Brain, are based on analysis of human brains from teenagers with recent head injury and mouse models that recreate sports-related head impact and military-related blast exposure. The investigators also performed laboratory experiments and computer modeling. Study results shed light on the origins of CTE and relationship to traumatic brain injury (TBI), concussion and subconcussive head injury.
CTE is a neurodegenerative disease characterized by abnormal accumulation of tau protein around small blood vessels in the brain. CTE causes brain cell death, cognitive deficits, and dementia. The brain pathology of CTE has been observed in brains of teenagers and adults with exposure to repeated head injury, both concussive and subconcussive episodes. However, the mechanisms that cause CTE and relationship to concussion, subconcussive injury and TBI remain poorly understood.
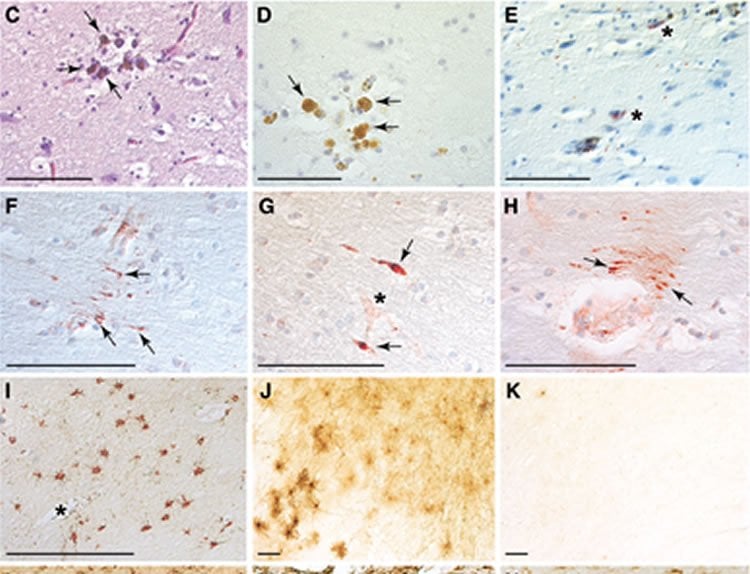
In the first part of their study, the researchers examined four postmortem brains from teenage athletes who had sustained closed-head impact injuries 1, 2, 10 and 128 days prior to death. Neuropathological analysis of these brains showed a spectrum of post-traumatic pathology, including one case of early-stage CTE and two cases with abnormal accumulation of tau protein. Brains from four age-matched athletes without recent head injury did not show the pathological changes observed in the head-injury group.
To investigate causal mechanisms underlying these changes, the researchers conducted laboratory experiments using mouse models of two different injury mechanisms–repeat closed-head impact and blast exposure–both linked to CTE. The investigators compared brain responses to the experimental injuries and relationship to CTE pathology over time.
Based on pathological findings in human cases, the researchers hypothesized that early CTE may result from damaged blood vessels in the brain that become leaky, resulting in blood proteins spilling into brain tissue and triggering brain inflammation. The researchers utilized a brain scan called dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) to detect leaky blood vessels in the brains of mice subjected to head impact. The investigators also found that head impact caused persistent changes in brain electrical functions, which may explain cognitive difficulties experienced by some people after these injuries.
“The same brain pathology that we observed in teenagers after head injury was also present in head-injured mice. We were surprised that the brain pathology was unrelated to signs of concussion, including altered arousal and impaired balance, among others. Our findings provide strong causal evidence linking head impact to TBI and early CTE, independent of concussion,” explained corresponding author Lee E. Goldstein, MD, PhD, an associate professor at Boston University School of Medicine and College of Engineering. “The results may explain why approximately 20 percent of athletes with CTE never suffered a diagnosed concussion.”
“Our experimental results showed no correlation between concussive signs at the time of injury and CTE brain pathology. These findings provide strong evidence–the best evidence we have so far–that subconcussive impacts are not only dangerous but also causally linked to CTE,” Goldstein said.
The unexpected findings led to the third part of the study, which utilized computer simulations performed at Lawrence Livermore National Laboratory, along with mechanical models implemented at Boston University. “The simulations showed that the mechanisms that trigger concussion may be distinct from those that lead to CTE,” said William C. Moss, PhD, physicist at Lawrence Livermore National Laboratory and co-author of the study. “Our results also provide fundamental insight into the origin of concussion.”
“In order to reduce CTE risk in contact sport athletes and military veterans, there must be a reduction in the number of head impacts. The continued focus on concussion and symptomatic recovery does not address the fundamental danger these activities pose to human health,” said Ann McKee, MD, chief of Neuropathology at Boston VA Healthcare System, Director of BU’s CTE Center and a co-author on this study.
“To prevent the disease, you have to prevent head impact – it’s hits to the head that cause CTE,” Goldstein added.
Goldstein and his colleagues anticipate that their study will advance understanding of how head injuries lead to CTE brain pathology, even in teenagers and young adults, with the goal being to facilitate development of new diagnostics, therapeutics, protective equipment and preventive measures to help people affected by head injuries and at risk for CTE.
Funding: The research was funded by the U.S. National Institutes of Health, U.S. Department of Veterans Affairs, U.S. Department of Defense, U.S. Department of Energy, Boston University Alzheimer’s Disease and CTE Center, University of California, Berkeley, WWE, National Football League, National Operating Committee on Standards for Athletic Equipment, European Union, Israel Science Foundation, Canadian Institute of Health Research, Andlinger Foundation and Concussion Legacy Foundation.
Source: Gina DiGravio – BMC
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to credited to Goldstein et al./Brain.
Original Research: Full open access research for “Concussion, microvascular injury, and early tauopathy in young athletes after impact head injury and an impact concussion mouse model ” by Chad A Tagge, Andrew M Fisher, Olga V Minaeva, Amanda Gaudreau-Balderrama, Juliet A Moncaster, Xiao-Lei Zhang, Mark W Wojnarowicz, Noel Casey, Haiyan Lu, Olga N Kokiko-Cochran, Sudad Saman, Maria Ericsson, Kristen D Onos, Ronel Veksler, Vladimir V Senatorov, Jr, Asami Kondo, Xiao Z Zhou, Omid Miry, Linnea R Vose, Katisha R Gopaul, Chirag Upreti, Christopher J Nowinski, Robert C Cantu, Victor E Alvarez, Audrey M Hildebrandt, Erich S Franz, Janusz Konrad, James A Hamilton, Ning Hua, Yorghos Tripodis, Andrew T Anderson, Gareth R Howell, Daniela Kaufer, Garth F Hall, Kun P Lu, Richard M Ransohoff, Robin O Cleveland, Neil W Kowall, Thor D Stein, Bruce T Lamb, Bertrand R Huber, William C Moss, Alon Friedman, Patric K Stanton, Ann C McKee, and Lee E Goldstein in Brain. Published online January 18 2018 doi:10.1093/brain/awx350
[cbtabs][cbtab title=”MLA”]BMC “Hits, Not Concussion, Cause CTE.” NeuroscienceNews. NeuroscienceNews, 18 January 2018.
<https://neurosciencenews.com/hits-cte-8335/>.[/cbtab][cbtab title=”APA”]BMC (2018, January 18). Hits, Not Concussion, Cause CTE. NeuroscienceNews. Retrieved January 18, 2018 from https://neurosciencenews.com/hits-cte-8335/[/cbtab][cbtab title=”Chicago”]BMC “Hits, Not Concussion, Cause CTE.” https://neurosciencenews.com/hits-cte-8335/ (accessed January 18, 2018).[/cbtab][/cbtabs]
Abstract
Concussion, microvascular injury, and early tauopathy in young athletes after impact head injury and an impact concussion mouse model
The mechanisms underpinning concussion, traumatic brain injury, and chronic traumatic encephalopathy, and the relationships between these disorders, are poorly understood. We examined post-mortem brains from teenage athletes in the acute-subacute period after mild closed-head impact injury and found astrocytosis, myelinated axonopathy, microvascular injury, perivascular neuroinflammation, and phosphorylated tau protein pathology. To investigate causal mechanisms, we developed a mouse model of lateral closed-head impact injury that uses momentum transfer to induce traumatic head acceleration. Unanaesthetized mice subjected to unilateral impact exhibited abrupt onset, transient course, and rapid resolution of a concussion-like syndrome characterized by altered arousal, contralateral hemiparesis, truncal ataxia, locomotor and balance impairments, and neurobehavioural deficits. Experimental impact injury was associated with axonopathy, blood–brain barrier disruption, astrocytosis, microgliosis (with activation of triggering receptor expressed on myeloid cells, TREM2), monocyte infiltration, and phosphorylated tauopathy in cerebral cortex ipsilateral and subjacent to impact. Phosphorylated tauopathy was detected in ipsilateral axons by 24 h, bilateral axons and soma by 2 weeks, and distant cortex bilaterally at 5.5 months post-injury. Impact pathologies co-localized with serum albumin extravasation in the brain that was diagnostically detectable in living mice by dynamic contrast-enhanced MRI. These pathologies were also accompanied by early, persistent, and bilateral impairment in axonal conduction velocity in the hippocampus and defective long-term potentiation of synaptic neurotransmission in the medial prefrontal cortex, brain regions distant from acute brain injury. Surprisingly, acute neurobehavioural deficits at the time of injury did not correlate with blood–brain barrier disruption, microgliosis, neuroinflammation, phosphorylated tauopathy, or electrophysiological dysfunction. Furthermore, concussion-like deficits were observed after impact injury, but not after blast exposure under experimental conditions matched for head kinematics. Computational modelling showed that impact injury generated focal point loading on the head and seven-fold greater peak shear stress in the brain compared to blast exposure. Moreover, intracerebral shear stress peaked before onset of gross head motion. By comparison, blast induced distributed force loading on the head and diffuse, lower magnitude shear stress in the brain. We conclude that force loading mechanics at the time of injury shape acute neurobehavioural responses, structural brain damage, and neuropathological sequelae triggered by neurotrauma. These results indicate that closed-head impact injuries, independent of concussive signs, can induce traumatic brain injury as well as early pathologies and functional sequelae associated with chronic traumatic encephalopathy. These results also shed light on the origins of concussion and relationship to traumatic brain injury and its aftermath.
“Concussion, microvascular injury, and early tauopathy in young athletes after impact head injury and an impact concussion mouse model ” by Chad A Tagge, Andrew M Fisher, Olga V Minaeva, Amanda Gaudreau-Balderrama, Juliet A Moncaster, Xiao-Lei Zhang, Mark W Wojnarowicz, Noel Casey, Haiyan Lu, Olga N Kokiko-Cochran, Sudad Saman, Maria Ericsson, Kristen D Onos, Ronel Veksler, Vladimir V Senatorov, Jr, Asami Kondo, Xiao Z Zhou, Omid Miry, Linnea R Vose, Katisha R Gopaul, Chirag Upreti, Christopher J Nowinski, Robert C Cantu, Victor E Alvarez, Audrey M Hildebrandt, Erich S Franz, Janusz Konrad, James A Hamilton, Ning Hua, Yorghos Tripodis, Andrew T Anderson, Gareth R Howell, Daniela Kaufer, Garth F Hall, Kun P Lu, Richard M Ransohoff, Robin O Cleveland, Neil W Kowall, Thor D Stein, Bruce T Lamb, Bertrand R Huber, William C Moss, Alon Friedman, Patric K Stanton, Ann C McKee, and Lee E Goldstein in Brain. Published online January 18 2018 doi:10.1093/brain/awx350







