Summary: Hearing loss associated with aging may have more to do with the death of hair cells in the ears than stria vascularis damage.
Source: SfN
Age-related hearing loss has more to do with the death of hair cells than the cellular battery powering them wearing out, according to new research in Journal of Neuroscience. That means wearing ear protection may prevent some age-related hearing loss.
Every day, people subject their ears and the delicate hair cells — the cells inside the cochlea that turn sound waves into electrical signals — within them to damaging noisy environments and too-loud headphones. However, ears also age. Both the hair cells and the stria vascularis, the cellular battery powering them, degrade with age. For 60 years, scientists attributed noise-induced hearing loss to hair cell death and age-related hearing loss to stria vascularis damage. But a new study from Wu et al. proves otherwise: age-related hearing loss in humans stems from hair cell death, not stria vascularis damage.
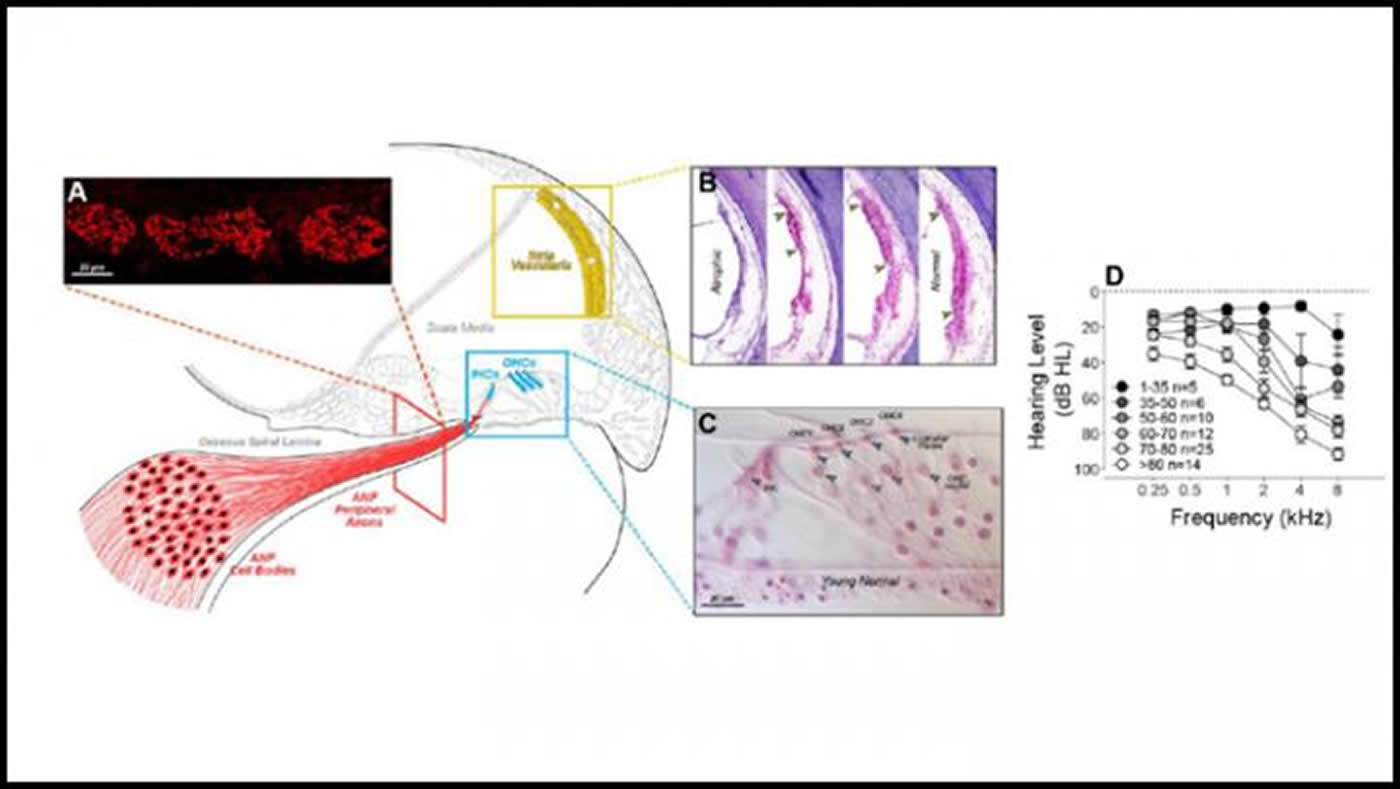
The research team counted surviving hair cells, auditory nerve fibers, and stria vascularis area in cochlea samples from 120 people and compared the damage to hearing test scores. Hair cell death predicted the severity of hearing loss, while stria vascularis damage did not. This contradicts findings in animal models, where the opposite is true. But animals do not experience the same auditory abuses as humans, which may mean that much of age-related hair cell loss is noise-induced, and therefore avoidable.
About this memory research article
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Wu et al., JNeurosci 2020.
Original Research: Closed access
“Age-Related Hearing Loss Is Dominated by Damage to Inner Ear Sensory Cells, Not the Cellular Battery That Powers Them”. by Pei-zhe Wu, Jennifer T. O’Malley, Victor de Gruttola and M. Charles Liberman. Journal of Neuroscience.
Abstract
Age-Related Hearing Loss Is Dominated by Damage to Inner Ear Sensory Cells, Not the Cellular Battery That Powers Them
Age-related hearing loss arises from irreversible damage in the inner ear, where sound is transduced into electrical signals. Prior human studies suggested that sensory-cell loss is rarely the cause; correspondingly, animal work has implicated the stria vascularis, the cellular “battery” driving the amplification of sound by hair cell “motors”. Here, quantitative microscopic analysis of hair cells, auditory nerve fibers and strial tissues in 120 human inner ears obtained at autopsy, most of whom had recent audiograms in their medical records, shows that the degree of hearing loss is well predicted from the amount of hair cell loss and that inclusion of strial damage does not improve the prediction. Although many aging ears showed significant strial degeneration throughout the cochlea, our statistical models suggest that, by the time strial tissues are lost, hair cell death is so extensive that the loss of battery is no longer important to pure-tone thresholds and that audiogram slope is not diagnostic for strial degeneration. These data comprise the first quantitative survey of hair cell death in normal-aging human cochleas, and reveal unexpectedly severe hair cell loss in low-frequency cochlear regions, and dramatically greater loss in high-frequency regions than seen in any aging animal model. Comparison of normal-aging ears to an age-matched group with acoustic-overexposure history suggests that a lifetime of acoustic overexposure is to blame.
Significance Statement
This report upends dogma about the causes of age-related hearing loss. Our analysis of over 120 autopsy specimens shows that inner-ear sensory cell loss can largely explain the audiometric patterns in aging, with minimal contribution from the stria vascularis, the “battery” that powers the inner ear, previously viewed as the major locus of age-related hearing dysfunction. Predicting inner ear damage from the audiogram is critical, now that clinical trials of therapeutics designed to regrow hair cells are underway. Our data also show that hair cell degeneration in aging humans is dramatically worse than that in aging animals, suggesting that the high-frequency hearing losses that define human presbycusis reflect avoidable contributions of chronic ear abuse to which aging animals are not exposed.






