Brain imaging explains reason for good and poor language outcomes in ASD toddlers.
Using functional magnetic resonance imaging (fMRI), University of California, San Diego School of Medicine researchers say it may be possible to predict future language development outcomes in toddlers with autistic spectrum disorder (ASD), even before they’ve been formally diagnosed with the condition.
The findings are published in the April 9 online issue of the journal Neuron.
A major challenge of ASD diagnosis and treatment is that the neurological condition – which affects 1 in 68 children in the United States, mostly boys – is considerably heterogeneous. Early symptoms differ between each ASD toddler, as does progression of the condition. No uniform clinical phenotype exists, in part because the underlying causes for different subtypes of autism are diverse and not well-understood.
“There is no better example than early language development,” said senior author Eric Courchesne, PhD, professor of neurosciences and co-director of the Autism Center of Excellence at UC San Diego. “Some individuals are minimally verbal throughout life. They display high levels of symptom severity and may have poor clinical outcomes. Others display delayed early language development, but then progressively acquire language skills and have relatively more positive clinical outcomes.”
In other words, said Courchesne, in some children with ASD language improves substantially with age; but in some it may progress too slowly or even diminish. The neurodevelopmental bases for this variability are unknown, he said. Differences in treatment quantity do not fully account for it. But numerous studies have shown that early, accurate diagnoses of ASD can improve treatment benefits in many affected children.
“It’s important to develop more and new biological ways to identify and stratify the ASD population into clinical sub-types so that we can create better, more individualized treatments,” said co-author Karen Pierce, PhD, associate professor of neurosciences and co-director of the Autism Center of Excellence.
In the Neuron paper, Courchesne, first author Michael V. Lombardo, PhD, a senior researcher at the University of Cambridge and assistant professor at the University of Cyprus, Pierce and colleagues describe the first effort to create a process capable of detecting different brain subtypes within ASD that underlie and help explain varying development language trajectories and outcomes. “We wanted to see if patterns of brain activity in response to language can explain and predict how well language skills would develop in a toddler with ASD before that toddler actually began talking,” said Courchesne.
The researchers combined prospective fMRI measurements of neural systems’ response to speech in children at the earliest ages at which risk of ASD can be clinically detected in a general pediatric population (at approximately ages 1-2 years) with comprehensive longitudinal diagnostic and clinical assessments of language skills at 3-4 years of age.
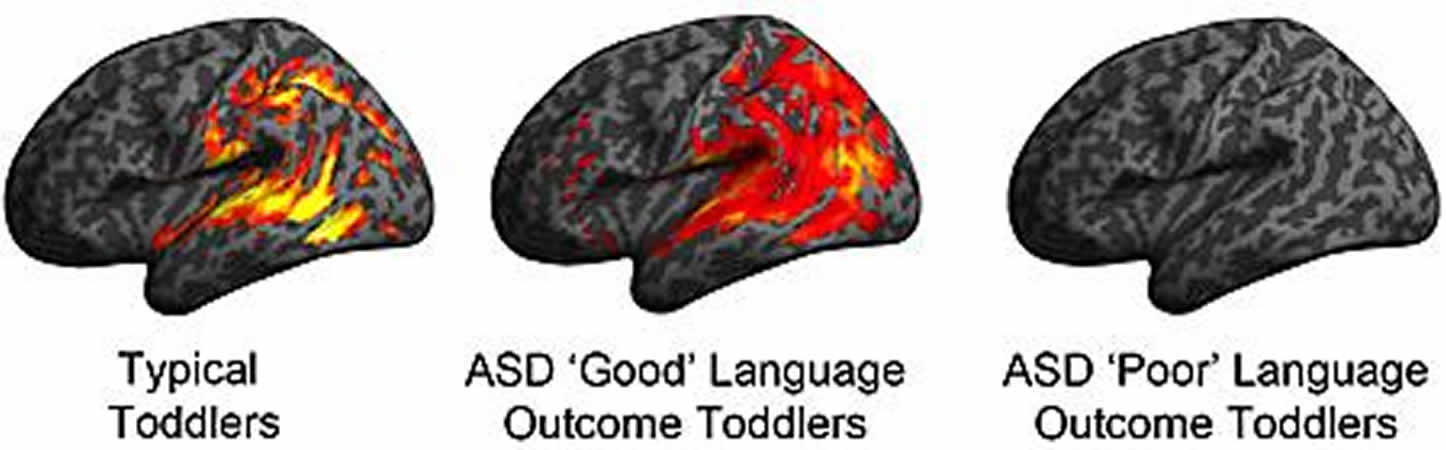
They found that pre-diagnosis fMRI response to speech in ASD toddlers with relatively good language outcomes was highly similar to non-ASD comparison groups with robust responses to language in superior temporal cortices, a region of the brain responsible for processing sounds so that they can be understood as language.
In contrast, ASD toddlers with poor language outcomes had superior temporal cortices that showed diminished or abnormal inactivity to speech.
In sum, the study found entirely different neural substrates at initial clinical detection that precede and underlie later good versus poor language outcome in autism. These findings, said researchers, will open new avenues of progress towards identifying the causes and best treatment for these two very different types of autism.
“For the first time, our study shows a strong relationship between irregularities in speech-activation in the language-critical superior temporal cortex and actual, real-world language ability in ASD toddlers,” said Lombardo.
The scientists said fMRI imaging also showed that the brains of ASD toddlers with poor language development processed speech differently, including how neural regions governing emotion, memory and motor skills were involved.
“Our work represents one of the first attempts using fMRI to define a neurofunctional biomarker of a subtype in very young ASD toddlers,” said Pierce. “Such subtypes help us understand the differences between persons with ASD. More importantly, they can help us determine how and why treatments are effective for some, but not all, on the autism spectrum.”
Co-authors include Lisa Eyler, UCSD and Veterans Affairs San Diego Healthcare System; Cindy Carter Barnes, Clelia Ahrens-Barbeau, Stephanie Solso, and Kathleen Campbell, UCSD.
Funding: Funding for this research came, in part, from National Institute of Mental Health grants P50-MH081755, R01-MH080134 and R01-MH036840, the National Foundation for Autism Research and Jesus College, Cambridge and the British Academy.
Written by Scott LaFee
Source: UCSD press release
Image Source: The image is supplied courtesy of UC San Diego School of Medicine
Original Research: Abstract for “Different Functional Neural Substrates for Good and Poor Language Outcome in Autism” by Michael V. Lombardo, Karen Pierce, Lisa T. Eyler, Cindy Carter Barnes, Clelia Ahrens-Barbeau, Stephanie Solso, Kathleen Campbell, and Eric Courchesne in Neuron. Published online April 9 2015 doi:10.1016/j.neuron.2015.03.023
Abstract
Different Functional Neural Substrates for Good and Poor Language Outcome in Autism
Highlights
•Different language trajectories emerge in ASD subgroups in the first 4 years of life
•ASD with later poor language outcome shows temporal cortex hypoactivation to speech
•Large-scale brain-language relationships are atypically reversed and specific to ASD
•ASD subgroup prognosis is best predicted by combining early behavioral and fMRI data
Summary
Autism (ASD) is vastly heterogeneous, particularly in early language development. While ASD language trajectories in the first years of life are highly unstable, by early childhood these trajectories stabilize and are predictive of longer-term outcome. Early neural substrates that predict/precede such outcomes are largely unknown, but could have considerable translational and clinical impact. Pre-diagnosis fMRI response to speech in ASD toddlers with relatively good language outcome was highly similar to non-ASD comparison groups and robustly recruited language-sensitive superior temporal cortices. In contrast, language-sensitive superior temporal cortices were hypoactive in ASD toddlers with poor language outcome. Brain-behavioral relationships were atypically reversed in ASD, and a multimodal combination of pre-diagnostic clinical behavioral measures and speech-related fMRI response showed the most promise as an ASD prognosis classifier. Thus, before ASD diagnoses and outcome become clinically clear, distinct functional neuroimaging phenotypes are already present that can shed insight on an ASD toddler’s later outcome.
“Different Functional Neural Substrates for Good and Poor Language Outcome in Autism” by Michael V. Lombardo, Karen Pierce, Lisa T. Eyler, Cindy Carter Barnes, Clelia Ahrens-Barbeau, Stephanie Solso, Kathleen Campbell, and Eric Courchesne in Neuron. Published online April 9 2015 doi:10.1016/j.neuron.2015.03.023






