Summary: A new study sheds light on why repeated concussions may contribute to the development of seizures.
Source: SfN.
Researchers have identified a cellular response to repeated concussions that may contribute to seizures in mice like those observed following traumatic brain injury in humans.
The study, published in Journal of Neuroscience, establishes a new animal model that could help improve our understanding of post-traumatic epilepsy.
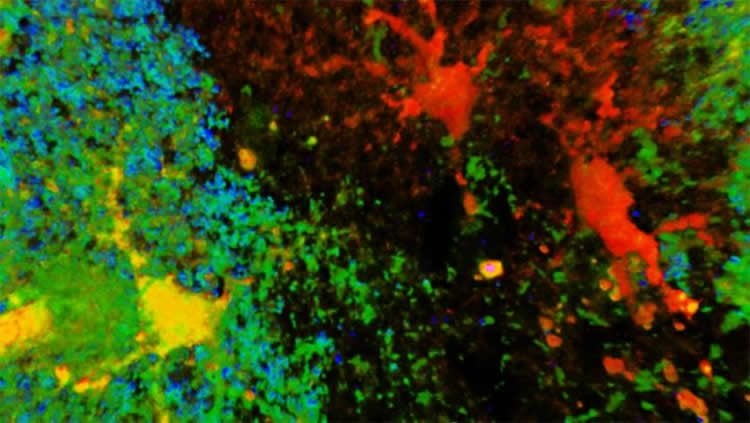
Stefanie Robel, Oleksii Shandra and colleagues induced mild traumatic brain injury in mice to mimic blows to the head that result in human concussions. A unique population of astrocytes responded to these injuries and the researchers observed spontaneous recurrent seizures in some mice within one month.
These results highlight the role of astrocytes in the development of epilepsy following the most common type of traumatic brain injury in humans.
Funding: NIH/National Institute of Neurological Disorders and Stroke, Institute for Critical Technology and Applied Science at Virginia Tech funded this study.
Source: David Barnstone – SfN
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Oleksii Shandra.
Original Research: Abstract for “Repetitive Diffuse Mild Traumatic Brain Injury Causes an Atypical Astrocyte Response and Spontaneous Recurrent Seizures” by Oleksii Shandra, Alexander R. Winemiller, Benjamin P. Heithoff, Carmen Munoz-Ballester, Kijana George, Michael J. Benko, Ivan Zuidhoek, Michelle N. Besser, Dallece E. Curley, G. Franklin Edwards, Anroux Mey, Alexys N. Harrington, Jeremy P. Kitchen and Stefanie Robel in Journal of Neuroscience. Published January 21 2019.
doi:10.1523/JNEUROSCI.1067-18.2018
[cbtabs][cbtab title=”MLA”]SfN”How Concussion May Lead to Epilepsy.” NeuroscienceNews. NeuroscienceNews, 21 January 2019.
<https://neurosciencenews.com/epilepsy-concussion-10595/>.[/cbtab][cbtab title=”APA”]SfN(2019, January 21). How Concussion May Lead to Epilepsy. NeuroscienceNews. Retrieved January 21, 2019 from https://neurosciencenews.com/epilepsy-concussion-10595/[/cbtab][cbtab title=”Chicago”]SfN”How Concussion May Lead to Epilepsy.” https://neurosciencenews.com/epilepsy-concussion-10595/ (accessed January 21, 2019).[/cbtab][/cbtabs]
Abstract
Repetitive Diffuse Mild Traumatic Brain Injury Causes an Atypical Astrocyte Response and Spontaneous Recurrent Seizures
Focal traumatic brain injury (TBI) induces astrogliosis, a process essential to protect uninjured brain areas from secondary damage. However, astrogliosis can cause loss of astrocyte homeostatic functions and possibly contributes to comorbidities such as post-traumatic epilepsy. Scar-forming astrocytes seal focal injuries off from healthy brain tissue. It is these glial scars that are associated with epilepsy originating in the cerebral cortex and hippocampus. Yet, the vast majority of human TBIs also present with diffuse brain injury caused by acceleration-deceleration forces leading to tissue shearing. The resulting diffuse tissue damage may be intrinsically different from focal lesions that would trigger glial scar formation. Here, we used mice of both sexes in a model of repetitive mild/concussive closed-head TBI, which only induced diffuse injury, to test the hypothesis that astrocytes respond uniquely to diffuse TBI and that diffuse TBI is sufficient to cause post-traumatic epilepsy. Astrocytes did not form scars and classic astrogliosis characterized by upregulation of glial fibrillary acidic protein was limited. Surprisingly, an unrelated population of atypical reactive astrocytes was characterized by the lack of glial fibrillary acidic protein expression, rapid and sustained downregulation of homeostatic proteins and impaired astrocyte coupling. After a latency period, a subset of mice developed spontaneous recurrent seizures reminiscent of post-traumatic epilepsy in human TBI patients. Seizing mice had larger areas of atypical astrocytes when compared to non-seizing mice suggesting that these atypical astrocytes might contribute to epileptogenesis after diffuse TBI.
SIGNIFICANCE STATEMENT
Traumatic brain injury (TBI) is a leading cause of acquired epilepsies. Reactive astrocytes have long been associated with seizures and epilepsy in patients, particularly after focal/lesional brain injury. However, most TBIs also include non-focal, diffuse injuries. Here, we showed that repetitive diffuse TBI is sufficient for the development of spontaneous recurrent seizures in a subset of mice. We identified an atypical response of astrocytes induced by diffuse TBI characterized by the rapid loss of homeostatic proteins and lack of astrocyte coupling while reactive astrocyte markers or glial scar formation was absent. Areas with atypical astrocytes were larger in animals that later developed seizures suggesting that this response may be one root cause of epileptogenesis after diffuse TBI.






