Glucose-enhanced MRI provides safer alternative for tumor imaging.
NIBIB-funded researchers have developed a method for imaging brain tumors that uses a common form of sugar. D-glucose, also known as dextrose, was used to enhance MRI images of brain tumors in three patients. The method takes advantage of the fact that growing tumors consume more sugar than normal tissues. This new approach offers a safer alternative to metal complexes, such as gadolinium that are commonly used to enhance MRI images but can have side effects in kidney patients and may build-up in the tissues of individuals needing multiple MRIs.
“This is the first non-metallic, biodegradable, natural MRI contrast agent tested in humans,” explained Guoying Liu, Ph.D., Director of the NIBIB Program in Magnetic Resonance Imaging and Spectroscopy. “Developing these natural contrast agents is critical for patients who require repeated imaging; additionally it will increase patient acceptance and comfort with imaging procedures.”
The work was performed by an international team including researchers at the Johns Hopkins University School of Medicine and Kennedy Krieger Institute in Baltimore, Maryland, and at Lund University in Lund, Sweden. The results are reported in the December issue of the journal TOMOGRAPHY.
Although MRI can be performed without contrast or enhancing agents, the addition of such agents is often critical for obtaining the detailed images needed for physicians to find the precise location of brain tumors.
The researchers performed a pilot study of Dynamic Glucose-Enhanced (DGE) MRI in four normal volunteers and three patients with brain tumors. When compared with MRI scans using the metallic enhancing agent gadolinium, DGE-MRI gave comparable results- showing details of brain structure and areas where tumors had disrupted the blood-brain barrier (BBB), which is a hallmark of the highest grade tumors.
“It is a significant step to be able to obtain clear MRI images of the brain using a biocompatible substance that is metabolized naturally by the body relatively quickly,” explained Peter van Zijl, Ph.D., Chief, Neuroscience, Division of MR Research at Johns Hopkins and senior author of the publication. “The dose of D-glucose is similar to that used for diabetes testing and is much cheaper than the metallic agents.”
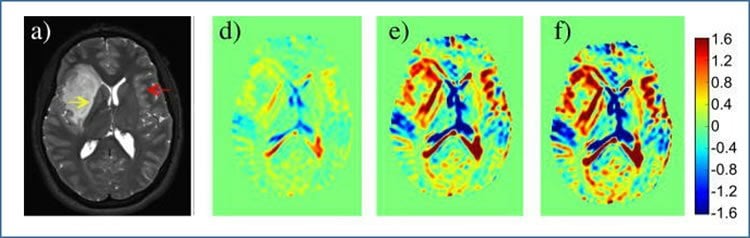
Dr. van Zijl explained that the glucose results are even more significant given very recent studies finding gadolinium from MRI procedures deposited in brain and bone. “These recent published findings are showing that gadolinium deposition in tissue could be more common than previously thought, so the development of a natural contrast agent alternative is certainly a high priority for the imaging community and, of course, patients.”
In addition, the use of glucose may offer more than a safer substitute for gadolinium. Because the highest grade tumors cause the most disruption of the BBB, the rate of movement of the contrast agent through the barrier is a strong indication that a tumor is high grade. Because gadolinium is larger than glucose it only detects 70 to 80 percent of high grade tumors–those with major disruption of the BBB, i.e. with holes large enough for gadolinium to penetrate. Because glucose is smaller it is expected to rapidly penetrate close to 100 percent of high grade tumors.
The next goal of van Zijl, with his collaborators in Sweden, is to test a larger group of patients to compare gadolinium enhancement to glucose enhancement. If glucose gives results comparable to gadolinium in a large group of patients, the team expects to quickly receive FDA approval for broad use of the technique.
Funding: The study was funded by the National Institutes of Health through grants EB019934 and EB018934 from the National Institute of Biomedical Imaging and Bioengineering, grant CA103175 from the National Cancer Institute, and grant DK093583 from the National Institute of Diabetes and Digestive and Kidney Disorders.
Source: Thomas Johnson – NIH/NIBIB
Image Credit: Image credited to Xu, et al./Tomography.
Original Research: Abstract for “Dynamic Glucose-Enhanced (DGE) MRI: Translation to Human Scanning and First Results in Glioma Patients” by Xiang Xu, Nirbhay N. Yadav, Linda Knutsson, Jun Hua, Rita Kalyani, Erica Hall, John Laterra, Jaishri Blakeley, Roy Strowd, Martin Pomper, Peter Barker, Kannie W. Y. Chan, Guanshu Liu, Michael T. McMahon, Robert D. Stevens, and Peter C.M. van Zijl in Tomography. Published online December 15 2015 doi:10.18383/j.tom.2015.00175
Abstract
Dynamic Glucose-Enhanced (DGE) MRI: Translation to Human Scanning and First Results in Glioma Patients
Recent animal studies have shown that D-glucose is a potential biodegradable magnetic resonance imaging (MRI) contrast agent for imaging glucose uptake in tumors. We show herein the first translation of that use of D-glucose to human studies. Chemical exchange saturation transfer (CEST) MRI at a single frequency offset optimized for detecting hydroxyl protons in D-glucose was used to image dynamic signal changes in the human brain at 7 T during and after D-glucose infusion. Dynamic glucose enhanced (DGE) image data from 4 normal volunteers and 3 glioma patients showed a strong signal enhancement in blood vessels, while a spa- tially varying enhancement was found in tumors. Areas of enhancement differed spatially between DGE and conventional gadolinium-enhanced imaging, suggesting complementary image information content for these 2 types of agents. In addition, different tumor areas enhanced with D-glucose at different times after infusion, suggesting a sensitivity to perfusion-related properties such as substrate delivery and blood-brain barrier (BBB) permeability. These preliminary results suggest that DGE MRI is feasible for studying glucose uptake in humans, providing a time-dependent set of data that contains information regarding arterial input function, tissue perfusion, glucose transport across the BBB and cell membrane, and glucose metabolism.
“Dynamic Glucose-Enhanced (DGE) MRI: Translation to Human Scanning and First Results in Glioma Patients” by Xiang Xu, Nirbhay N. Yadav, Linda Knutsson, Jun Hua, Rita Kalyani, Erica Hall, John Laterra, Jaishri Blakeley, Roy Strowd, Martin Pomper, Peter Barker, Kannie W. Y. Chan, Guanshu Liu, Michael T. McMahon, Robert D. Stevens, and Peter C.M. van Zijl in Tomography. Published online December 15 2015 doi:10.18383/j.tom.2015.00175






