Summary: A new study sheds light on the mechanisms of gyrus formation.
Source: Kanazawa University.
The human brain is highly developed with higher functions compared to other animals, thanks to which humans are believed to have acquired special abilities. One of the important factors considered to contribute to the development of the brain is the gyrus, the fold on the cerebral cortex surface.
The cerebral cortex is particularly important in higher brain functions. The cerebral cortex of higher animals, including humans, has many folds, called the gyrus (plural: “gyri”). By acquiring the gyrus in the process of evolution, it has become possible to have a large number of neurons, and thus great development of brain functions has been acquired. On the other hand, the mouse, a widely used model animal, has a brain without gyri. This has made it very difficult to do research on the gyrus using the mouse as a model animal; therefore, research on the gyrus has been much retarded.
Our research group has been using the ferret, a higher mammal, that has a brain with a gyrus, rather similar to the human brain. Our group has been developing a research technique for ferrets reported in several articles, which allows analysis of ferrets at the genome level. Our laboratory is a world leader in research in this field. Furthermore, by using this technique, we have succeeded in developing a ferret disease model that shows impairment in the gyrus, which, we believe, has significantly contributed to genetic approaches to the brain of higher mammals.
Results
By using the latest genome editing tool, a type of genetic engineering, we have succeeded in knocking out a specific gene in the ferret cerebral cortex and in elucidating one of the mechanisms of gyrus formation.
- Establishment of a technique for knocking out a specific gene in the ferret cerebral cortex by using the latest genome editing tool:
We have succeeded in establishing a technique for knocking out any gene in the ferret cerebral cortex by combining CRISPR/Cas9, the latest genome editing tool, and the technique we have so far developed for introducing a gene in the ferret cerebral cortex. This new technique opens a way to a new stage of genetics research of the brain of higher mammals. - Important neurons for gyrus formation:
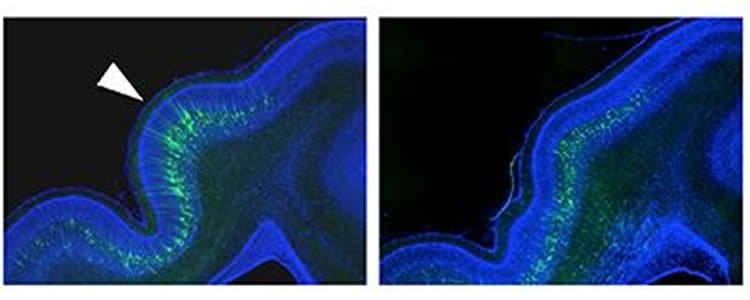
We searched for neurons playing important roles in gyrus formation and found that neurons on the upper-layer cerebral cortex were essential. This finding is the first in the world; it was not known so far which neurons in the brain would play important roles in gyrus formation.
We have applied the above-mentioned technique to knocking out a gene called Cdk5 in the ferret cerebral cortex and found that gyrus formation was impaired. This result indicates that Cdk5 is an important gene for gyrus formation.
These results of the current study indicate that functions of Cdk5 in neurons of the upper-layer cerebral cortex are important for the formation of the gyrus.
Significance and future prospect
In this study, our research group has elucidated one of the mechanisms for gyrus formation by developing a research technique for the ferret brain. Until today, research on gyrus formation has been rare so this study is truly world-leading. It is expected that this study will pave a way to elucidate brain evolution, up to the human brain, which was previously quite difficult with mice, a widely used model animal.
In addition, humans suffer, although rarely, from diseases due to gyrus impairment, the pathogenesis of which is largely unknown. The new technique we have developed in this study is expected to lead to elucidation of the etiology of various diseases including those mentioned above, which was difficult to investigate in the mouse.
Funding: The research was funded by the Japan Society for the Promotion of Science, Takeda Science Foundation, Uehara Memorial Foundation, Inamori Foundation, Senri Life Science Foundation.
Source: Fujiko Imanaga – Kanazawa University
Image Source: NeuroscienceNews.com image is credited to Kanazawa University.
Original Research: Full open access research for “Folding of the Cerebral Cortex Requires Cdk5 in Upper-Layer Neurons in Gyrencephalic Mammals” by Yohei Shinmyo, Yukari Terashita, Tung Anh Dinh Duong, Toshihide Horiike, Muneo Kawasumi, Kazuyoshi Hosomichi, Atsushi Tajima, and Hiroshi Kawasaki in Cell Reports. Published online August 29 2017 doi:10.1016/j.celrep.2017.08.024
[cbtabs][cbtab title=”MLA”]Kanazawa University “Folding of the Cerebral Cortex: Identification of Important Neurons.” NeuroscienceNews. NeuroscienceNews, 5 October 2017.
<https://neurosciencenews.com/cerebral-cortex-folding-7664/>.[/cbtab][cbtab title=”APA”]Kanazawa University (2017, October 5). Folding of the Cerebral Cortex: Identification of Important Neurons. NeuroscienceNews. Retrieved October 5, 2017 from https://neurosciencenews.com/cerebral-cortex-folding-7664/[/cbtab][cbtab title=”Chicago”]Kanazawa University “Folding of the Cerebral Cortex: Identification of Important Neurons.” https://neurosciencenews.com/cerebral-cortex-folding-7664/ (accessed October 5, 2017).[/cbtab][/cbtabs]
Abstract
Folding of the Cerebral Cortex Requires Cdk5 in Upper-Layer Neurons in Gyrencephalic Mammals
Highlights
•Efficient gene knockout in the ferret cerebral cortex was achieved using CRISPR/Cas9
•Cdk5 knockout in the ferret cerebral cortex markedly impairs cortical folding
•Appropriate positioning of upper-layer neurons is critical for cortical folding
Summary
Folds in the cerebral cortex in mammals are believed to be key structures for accommodating increased cortical neurons in the cranial cavity. However, the mechanisms underlying cortical folding remain largely unknown, mainly because genetic manipulations for the gyrencephalic brain have been unavailable. By combining in utero electroporation and the CRISPR/Cas9 system, we succeeded in efficient gene knockout of Cdk5, which is mutated in some patients with classical lissencephaly, in the gyrencephalic brains of ferrets. We show that Cdk5 knockout in the ferret cerebral cortex markedly impaired cortical folding. Furthermore, the results obtained from the introduction of dominant-negative Cdk5 into specific cortical layers suggest that Cdk5 function in upper-layer neurons is more important for cortical folding than that in lower-layer neurons. Cdk5 inhibition induced severe migration defects in cortical neurons. Taken together, our findings suggest that the appropriate positioning of upper-layer neurons is critical for cortical folding.
“Folding of the Cerebral Cortex Requires Cdk5 in Upper-Layer Neurons in Gyrencephalic Mammals” by Yohei Shinmyo, Yukari Terashita, Tung Anh Dinh Duong, Toshihide Horiike, Muneo Kawasumi, Kazuyoshi Hosomichi, Atsushi Tajima, and Hiroshi Kawasaki in Cell Reports. Published online August 29 2017 doi:10.1016/j.celrep.2017.08.024






