Summary: When the brain is deprived of input from a limb following amputation, it reorganizes its neural map and reroutes the function to the remaining limb.
Source: University of Missouri-Columbia
Fans of the blockbuster movie “Iron Man 3” might remember the characters step inside the digital projection of a “big brain” and watch as groups of neurons are “lit up” along the brain’s neural “map” in response to physical touch. Now, much like that scene, researchers at the University of Missouri have discovered a new insight into how the complex neural map of the human brain operates. Similar findings have been previously reported in animal studies, but this is one of the first studies where such a result has been documented in people.
“When a person touches something with their right hand, a specific ‘hand area’ in the left side of the brain lights up,” said Scott Frey, the Miller Family Chair in Cognitive Neuroscience in the Department of Psychological Sciences. “A similar, but opposite reaction happens with the left hand. But when someone loses a hand, we found both ‘hand areas’ of the brain — left and right — become dedicated to the remaining healthy hand. This is a striking example of functional reorganization or the plasticity of the human brain.”
Researchers used functional MRI (fMRI) at the MU Brain Imaging Center to scan the brains of 48 people — 19 of whom had lost a hand. They created a computer-controlled, air-based system to deliver light touch to the hands and face. Functional MRI scans are similar to traditional MRI scans but are sensitive to tiny changes in blood oxygenation levels in the brain that occur when areas of the brain are processing information. The researchers saw in their scans that when the brain is deprived of input from a lost hand, it reorganizes its neural map and reroutes those functions to the remaining hand.
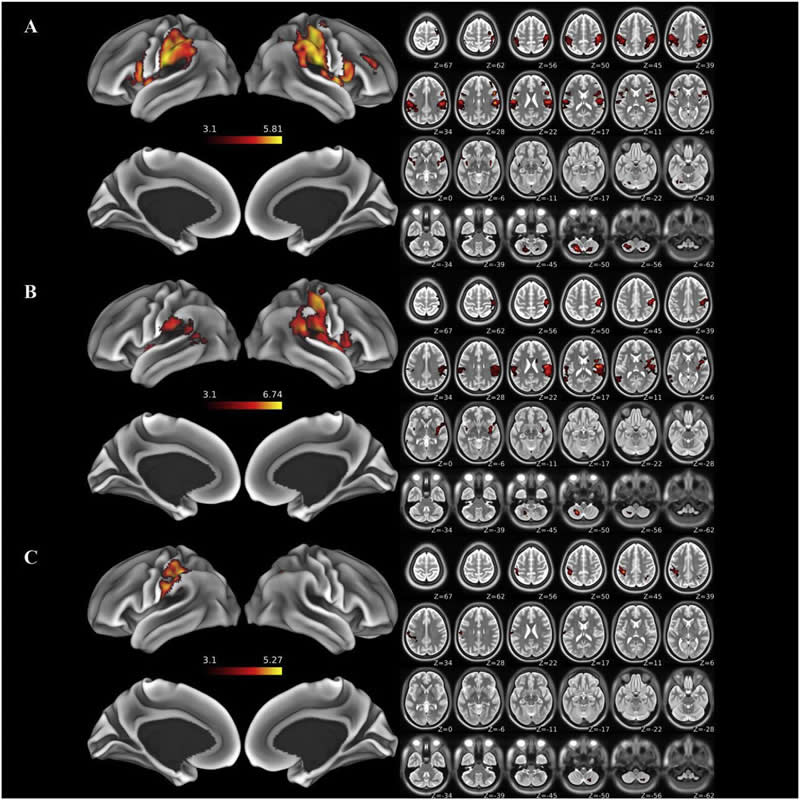
Frey said this discovery could help scientists and medical professionals better understand the underlying mechanisms behind the brain’s plasticity — the ability for the brain to adapt to changing conditions — when a traumatic bodily injury occurs, such as with veterans returning from injury on the military battlefield.
“We can think of the areas of the brain that process sensations from our bodies as being organized like a map with separate territories devoted to specific body regions such as the hands, face, or feet,” said Frey, who is also the director of the Rehabilitation Neuroscience Laboratory at MU, a joint venture between the MU College of Arts and Science and the MU School of Medicine. “We have long known that injuries such amputation or spinal cord damage change the organization of this map. If you lose a hand, for instance, then the associated ‘hand area’ may be partially taken over by neighboring functions in the map involved in processing sensations of the arm or face. This is a form of ‘brain plasticity.’ This work demonstrates that such plasticity also occurs across great distances between the left and right hemispheres of the brain.”
Researchers said additional work is underway to determine how and whether these changes impact how amputees experience sensations, including pain. The scientists hope their findings might also help inform efforts to develop prostheses that can provide users with the experience of touch.
Funding: Funding was provided by a grant from the National Institutes of Health and a grant from the U.S. Department of Defense. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies.
Source:
University of Missouri-Columbia
Media Contacts:
Eric Stann – University of Missouri-Columbia
Image Source:
The image is credited to University of Missouri.
Original Research: Closed access
“Interhemispheric transfer of post-amputation cortical plasticity within the human somatosensory cortex”. Scott Frey et al.
NeuroImage doi:10.1016/j.neuroimage.2019.116291.
Abstract
Interhemispheric transfer of post-amputation cortical plasticity within the human somatosensory cortex
Animal models reveal that deafferenting forelimb injuries precipitate reorganization in both contralateral and ipsilateral somatosensory cortices. The functional significance and duration of these effects are unknown, and it is unclear whether they also occur in injured humans. We delivered cutaneous stimulation during functional magnetic resonance imaging (fMRI) to map the sensory cortical representation of the intact hand and lower face in a group of chronic, unilateral, upper extremity amputees (N = 19) and healthy matched controls (N = 29). Amputees exhibited greater activity than controls within the deafferented former sensory hand territory (S1f) during stimulation of the intact hand, but not of the lower face. Despite this cortical reorganization, amputees did not differ from controls in tactile acuity on their intact hands. S1f responses during hand stimulation were unrelated to tactile acuity, pain, prosthesis usage, or time since amputation. These effects appeared specific to the deafferented somatosensory modality, as fMRI visual mapping paradigm failed to detect any differences between groups. We conclude that S1f becomes responsive to cutaneous stimulation of the intact hand of amputees, and that this modality-specific reorganizational change persists for many years, if not indefinitely. The functional relevance of these changes, if any, remains unknown.






