Summary: A new study has unveiled the first comprehensive neural blueprint explaining how fruit flies (Drosophila melanogaster) execute lightning-fast escape behaviors. By mining a high-resolution electron microscopy “connectome” of the fly’s ventral nerve cord (the insect equivalent of a spinal cord), researchers mapped all 1,314 descending neurons.
They discovered that rare, highly selective neuron-to-neuron connections called axo-axonic synapses act as powerful modulators that boost and synchronize motor commands, offering a decentralized and resilient framework for rapid decision-making.
Key Facts
- Mapping the Ventral Cord: Researchers analyzed all 1,314 descending neurons—cells that carry commands from the brain to the body—to identify instances of axo-axonic connectivity.
- The Power of Axo-Axonic Synapses: Unlike standard synapses, axo-axonic connections allow one axon to directly influence another axon before the signal ever reaches the muscles, enabling rapid signal modulation.
- Extraordinary Selectivity: These specialized connections are remarkably rare, forming in only about 1% of all possible neuron pairings within the motor circuitry.
- Decentralized “Broker” Network: Instead of relying on a few dominant “superhub” neurons, the fly’s escape network uses a distributed architecture of interconnected “broker” neurons, eliminating single points of failure.
- Amplifying Giant Fibers: The study demonstrated that specific axo-axonic neurons directly amplify “giant fibers”, the primary escape-command neurons, increasing the probability of a split-second getaway.
Source: FAU
Have you ever wondered how a fly manages to dodge you in a split second? Scientists have long been fascinated by the lightning-fast reflexes that help flies escape danger almost instantly.
But despite decades of research, they still don’t fully understand exactly how the brain coordinates these rapid reactions at the level of individual neural connections.
Now, a new Florida Atlantic University study offers the first comprehensive blueprint of a specialized neural wiring system linked to these escape behaviors in the fruit fly (Drosophila melanogaster).
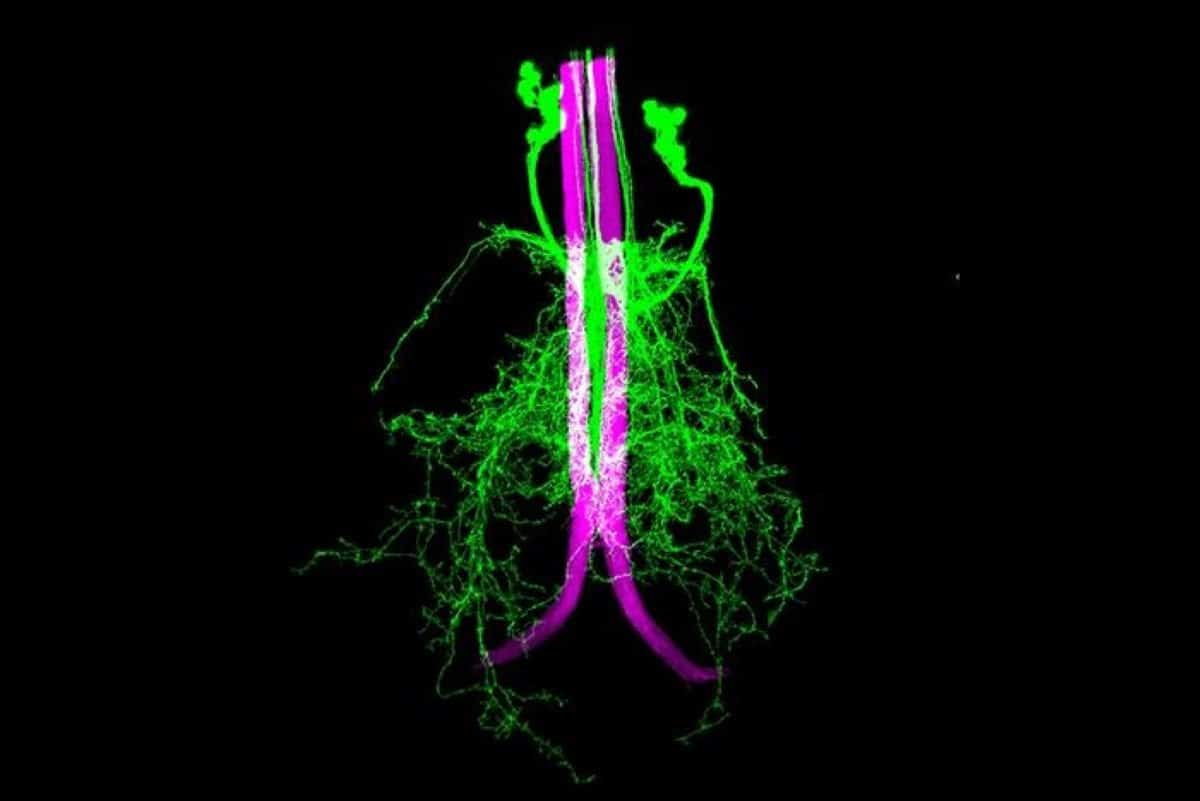
Using one of the most detailed maps ever created of the fly nervous system, researchers uncovered how rare neuron-to-neuron connections called axo-axonic synapses help fine-tune the rapid signals that drive split-second escape responses.
The findings, published in iScience, a Cell Press journal, provide new insight into how brains process information at extraordinary speed, bridging a critical gap between neural wiring and motor function, and offering a foundation for next-generation models of rapid decision-making in both invertebrates and vertebrates.
Using one of the most detailed neural maps ever assembled, FAU researchers analyzed all 1,314 descending neurons – brain-originating nerve cells that transmit commands from the brain to the body – within the fruit fly’s ventral nerve cord, the insect equivalent of a spinal cord.
The team mined a complete electron microscopy “connectome,” a high-resolution wiring diagram of the nervous system, to identify every instance of axo-axonic connectivity, a specialized form of neuron-to-neuron communication in which one axon directly influences another axon before signals reach muscles or other target cells.
“Our findings reveal a previously hidden wiring logic for how nervous systems achieve rapid and reliable motor control,” said Rodrigo Pena, Ph.D., senior author, an assistant professor of biological sciences, within FAU’s Charles E. Schmidt College of Science on the John D. MacArthur Campus in Jupiter, and a member of the FAU Stiles-Nicholson Brain Institute.
“What is especially exciting is that we uncovered a decentralized communication strategy that appears both highly efficient and remarkably robust. These principles may represent a conserved blueprint shared across species, from insects to vertebrates, and could ultimately help us better understand how brains coordinate fast decisions, movement and survival behaviors.”
The researchers combined large-scale computational modeling, network analysis and live optogenetic experiments – using light to activate specific neurons – to determine how these rare connections shape rapid motor responses such as escape behaviors. Their analysis revealed that axo-axonic connections are extraordinarily selective, forming in only about 1% of all possible neuron pairings.
“Despite their rarity, the network creates a highly efficient communication system in which signals can rapidly spread across the motor circuitry in only a few steps,” said Pena.
The study also found that the fly’s motor control network operates differently from many other known brain systems. Rather than relying on a few dominant “superhub” neurons, control is distributed across many interconnected “broker” neurons, creating a decentralized architecture that is both flexible and resilient. This arrangement may allow flies to rapidly combine reflexive movements with coordinated whole-body actions while avoiding single points of failure.
Importantly, the researchers demonstrated that specific axo-axonic neurons can directly amplify escape-command neurons known as giant fibers, increasing the likelihood that rapid escape signals will fire. Axo-axonic neurons are difficult to find and study in mammals, but these results are interesting because they can explain the importance of this unusual type of connection.
The findings suggest that these specialized synapses act as powerful modulators capable of boosting, suppressing or synchronizing motor commands before movement even begins.
“This study gave us an unprecedented opportunity to explore neural communication at a level of detail that simply wasn’t possible before,” said César C. Ceballos, Ph.D., first author, a postdoctoral fellow in the Charles E. Schmidt College of Science, and a member of the FAU Stiles-Nicholson Brain Institute.
“To discover that such sparse connections can still create a system-wide network capable of influencing behavior so rapidly was incredibly surprising. It suggests these hidden circuits may be far more influential in driving rapid responses than previously understood.”
The study involved an interdisciplinary team of researchers from three laboratories on FAU’s Jupiter campus. Study co-authors are Juan Lopez, Ph.D., a postdoctoral researcher of computational neuroscience at FAU; Ty Roachford, a neuroscience Ph.D. student in the Pena lab at FAU; Casey L. Spencer, Ph.D., an assistant professor of neuroscience in FAU’s Harriet L. Wilkes Honors College; and Rodney Murphey, Ph.D., a professor of biological sciences in the Charles E. Schmidt College of Science.
Key Questions Answered:
A: It comes down to a specialized, ultra-fast bypass in their wiring. Flies have rare connections called axo-axonic synapses that let nerve cells talk directly to each other “axon-to-axon” right before the signal hits the muscles. This cuts out intermediate steps, allowing signals to flood the motor system almost instantly.
A: If a network relies on one or two “superhub” neurons, injuring those cells destroys the entire system. By distributing control across many interconnected “broker” neurons, the fly’s brain creates a decentralized architecture. This makes their reflexes incredibly flexible, robust, and completely free of a single point of failure.
A: Yes. Axo-axonic connections exist in mammals, but they are notoriously difficult to find and study in larger brains. Because these basic motor control principles are highly efficient, scientists believe they represent a conserved evolutionary blueprint shared across species, which could help us model rapid human decisions and survival behaviors.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this neuroscience research news
Author: Gisele Galoustian
Source: FAU
Contact: Gisele Galoustian – FAU
Image: The image is credited to Casey Spencer, Ph.D., Florida Atlantic University
Original Research: Open access.
“The Drosophila connectome reveals axo-axonic synapses on descending neurons” by César Ceballos, Juan Lopez, Ty Roachford, Daniel Sanchez, Sabrina Jara, Kelli Robbins, Casey L. Spencer, Rodney Murphey, and Rodrigo F.O. Pena. iScience
DOI:10.1016/j.isci.2026.115624
Abstract
The Drosophila connectome reveals axo-axonic synapses on descending neurons
Axo-axonic synapses can veto, amplify, or synchronize spikes, yet their circuit-scale logic is unknown.
Using the complete electron microscopy connectome of the adult male Drosophila, we charted every axo-axonic input onto the 1,314 descending neurons that carry brain commands to the ventral nerve cord.
By definition, any synapse connected to a descending neuron within the cord is axo-axonic.
Thus, we uncovered the ascending-descending and interneurons-descending axo-axonic relationship.
Neurons with many partners (high-degree) integrate into the network without clustering into an interconnected “rich-club” of hubs.
We identified an octet of ascending neurons (AN08B098) whose axo-axonic input to the giant fibers (DNp01) predicted modulation of the escape circuit. Immunostaining confirms their cholinergic identity, while optogenetic activation confirmed that this excitatory cohort increases DNp01 excitability, validating connectome-derived rules.
Our work delivers a map of axo-axonic wiring in a complete ventral nerve cord connectome and provides constraints for models of fast motor control.






