Summary: Combining focused ultrasound with microbubble treatments weakens the connection between blood-brain barrier cells, effectively allowing for better delivery and absorption of drug treatments.
Source: QIMR Berghofer Medical Research Institute
A new QIMR Berghofer study has added weight to the potential benefits of using ultrasound treatment to deliver disease-targeting drugs to Alzheimer’s patients.
The study is the first to examine the technique on brain cells derived from human patients with Alzheimer’s disease, building on previous research on mice and other animal models.
The findings have been published today in the journal Stem Cell Reports.
Lead Australian researcher and head of QIMR Berghofer’s Cellular and Molecular Neurodegeneration group, Associate Professor Anthony White, said the researchers found using focused ultrasound coupled with microbubble treatment, could create openings in the blood-brain barrier formed by human endothelial cells.
“The blood-brain barrier is a semipermeable barrier that lines blood vessels in the brain and importantly protects brain tissue, but that protective function also prevents the uptake of drugs and therapies targeting brain diseases,” Associate Professor White said.
“We found by using focused ultrasound and microbubble treatment we could weaken the connection between blood-brain barrier cells, which would potentially allow the brain tissue to absorb a drug treatment.
“An abnormal blood-brain barrier has been associated with several neurodegenerative diseases, including Alzheimer’s disease, so it’s very important that we better understand how the barrier works and how we can safely penetrate it to combat disease.
“Our study is the first to look at how the blood-brain barrier cells from human patients can be disrupted to improve the uptake of Alzheimer’s therapies, building on previous studies that have explored if ultrasound could be used to reduce amyloid build up in the brains of mice and other animal models.”
The researchers used brain endothelial cells derived from human stem cells of people with a family history of Alzheimer’s disease to understand how those cells of the blood-brain barrier could be weakened.
They injected lipid microbubbles into the cells and then targeted the area with ultrasound which caused the cells to expand and contract, disrupting the connections between the cells, opening the blood-brain barrier. The ultrasound and microbubble treatment technique was developed by researchers at the Queensland Brain Institute.
First author and QIMR Berghofer researcher, Dr. Lotta Oikari, said the study also showed the ultrasound and microbubble treatment had a longer lasting effect on the brain cells of Alzheimer’s patients than on healthy controls.
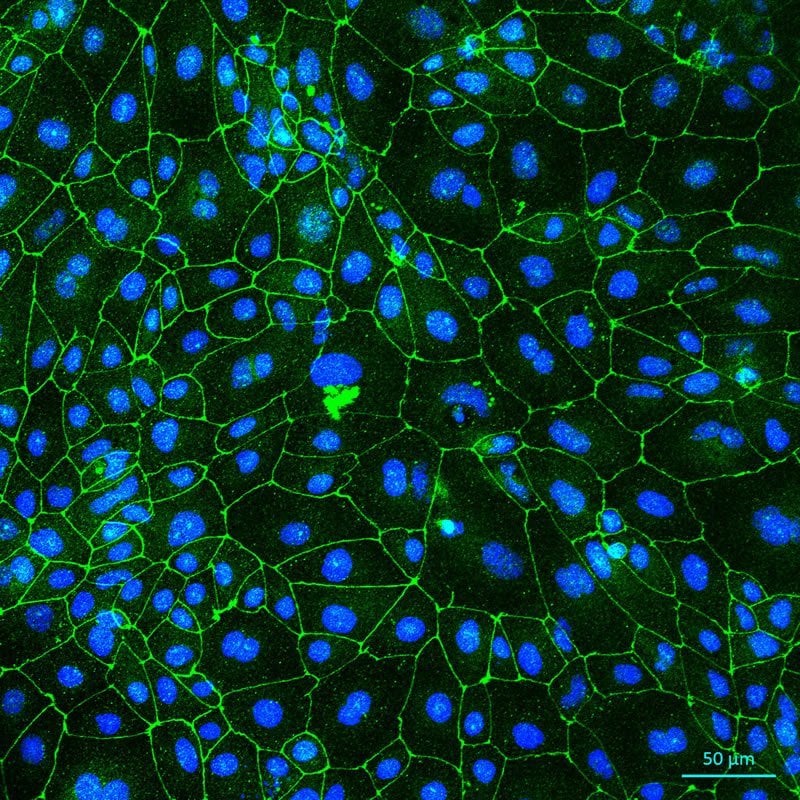
“The treatment generated openings in the monolayer of the blood-brain barrier of all patients, but the brain endothelial cells of healthy controls repaired themselves quicker than the Alzheimer’s patient cells,” Dr. Oikari said.
“The blood-brain barrier in Alzheimer’s patients was slower to repair, indicating they would be more receptive to drugs and treatments for longer and that brain ultrasound treatment may have to be adjusted differently depending on the type of disease the patient has.
“It also raises questions whether reduced integrity in the blood-brain barrier, following ultrasound treatment, could lead to lower amyloid levels in the brains of Alzheimer’s patients because the brain tissue could potentially expel the build-up.”
Funding: The research was funded by QIMR Berghofer.
The study involved researchers from QIMR Berghofer, the Queensland Brain Institute at The University of Queensland, Monash University, University of Helsinki and University of Eastern Finland, in Finland.
About this neurology research article
Source:
QIMR Berghofer Medical Research Institute
Media Contacts:
Press Office – QIMR Berghofer Medical Research Institute
Image Source:
The image is credited to Lotta Oikari.
Original Research: Open access
“Altered Brain Endothelial Cell Phenotype from a Familial Alzheimer Mutation and Its Potential Implications for Amyloid Clearance and Drug Delivery”. Lotta E. Oikari et al.
Stem Cell Reports doi:10.1016/j.stemcr.2020.03.011.
Abstract
Altered Brain Endothelial Cell Phenotype from a Familial Alzheimer Mutation and Its Potential Implications for Amyloid Clearance and Drug Delivery
The blood-brain barrier (BBB) presents a barrier for circulating factors, but simultaneously challenges drug delivery. How the BBB is altered in Alzheimer disease (AD) is not fully understood. To facilitate this analysis, we derived brain endothelial cells (iBECs) from human induced pluripotent stem cells (hiPSCs) of several patients carrying the familial AD PSEN1 mutation. We demonstrate that, compared with isogenic PSEN1 corrected and control iBECs, AD-iBECs exhibit altered tight and adherens junction protein expression as well as
efflux properties. Furthermore, by applying focused ultrasound (FUS) that transiently opens the BBB and achieves multiple therapeutic effects in AD mouse models, we found an altered permeability to 3–5 kDa dextran as a model cargo and the amyloid-b (Ab) peptide in ADiBECs compared with control iBECs. This presents human-derived in vitro models of the BBB as a valuable tool to understand its role and properties in a disease context, with possible implications for drug delivery.
Feel Free To Share This Alzheimer’s Disease News.






