Summary: Researchers report activating a pathway between the amygdala and brain stem helps to relieve pain and reduce defensive behaviors in rat models.
Source: SfN
Activating a circuit between the amygdala and brainstem relieves pain and reduces defensive behaviors in rats, according to research recently published in Journal of Neuroscience.
People with pain often experience psychological comorbidities like anxiety and depression, which can end up making their prognosis worse.
A pathway from the amygdala to a nucleus in the brainstem may offer a way to treat pain’s toll on both the body and the mind.
Hogri et al. activated the circuit in rats and measured their response to chemical, mechanical, and thermal pain stimuli. Stimulating the circuit decreased the brainstem’s response to all three stimuli, indicating pain relief.
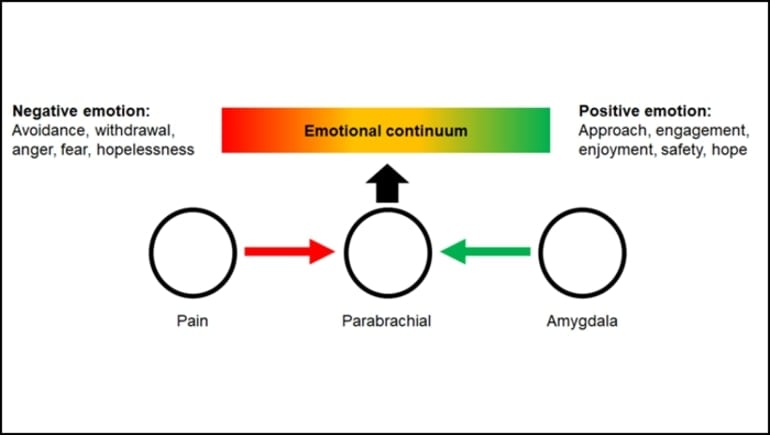
In a separate situation, stimulating the circuit decreased the rats’ defensive behaviors in response to a threat. It also increased reward and feeding behavior, a sign the rats felt safe.
The combination of these behavior changes indicates the circuit steers rats out of a negative emotional state and into a positive one.
In humans, there is not yet a way to stimulate a specific brain circuit.
Existing brain stimulation technologies are not precise enough and could activate neighboring circuits that could increase pain and negative emotions.
But addressing these technical roadblocks could lead to a treatment that addresses both the pain itself and the negative feelings it generates.
About this pain and emotion research news
Author: Calli McMurray
Source: SfN
Contact: Calli McMurray – SfN
Image: The image is credited to Roni Hogri, 2022.
Original Research: Closed access.
“GABAergic CaMKIIα+ amygdala output attenuates pain and modulates emotional-motivational behaviour via parabrachial inhibition” by Roni Hogri, et al. Journal of Neuroscience
Abstract
GABAergic CaMKIIα+ amygdala output attenuates pain and modulates emotional-motivational behaviour via parabrachial inhibition
Pain and emotion are strongly regulated by neurons in the central nucleus of the amygdala (CeA), a major output of the limbic system; yet, the neuronal signalling pathways underlying this modulation are incompletely understood.
Here, we characterized a subpopulation of CeA neurons that express the CaMKIIα gene (CeACAM neurons) and project to the lateral parabrachial nucleus (LPBN), a brainstem region known for its critical role in distributing nociceptive and other aversive signals throughout the brain.
In male Sprague-Dawley rats, we show that CeACAM-LPBN neurons are GABAergic and mostly express somatostatin. In anaesthetised rats, optogenetic stimulation of CeACAM-LPBN projections inhibited responses of LPBN neurons evoked by electrical activation of Aδ- and C-fibre primary afferents; this inhibition could be blocked by intra-LPBN application of the GABAA receptor antagonist bicuculline. CeACAM-LPBN stimulation also dampened LPBN responses to noxious mechanical, thermal, and chemical stimuli.
In behaving rats, optogenetic stimulation of CeACAM-LPBN projections attenuated nocifensive responses to mechanical pressure and radiant heat, disrupted the ability of a noxious shock to drive aversive learning, reduced the defensive behaviours of thigmotaxis and freezing, induced place preference, and promoted food consumption in sated rats.
Thus, we suggest that CeACAM-LPBN projections mediate a form of analgesia that is accompanied by a shift towards the positive-appetitive pole of the emotional-motivational continuum.
Since the affective state of pain patients strongly influences their prognosis, we envision that recruitment of this pathway in a clinical setting could potentially promote pain resilience and recovery.