Discovery opens door to development of new drugs to control weight gain and obesity.
It’s rare for scientists to get what they describe as “clean” results without spending a lot of time repeating the same experiment over and over again. But when researchers saw the mice they were working with doubling their weight within a month or two, they knew they were on to something.
“About twenty years ago there was a big step forward in our understanding of obesity when researchers discovered that our appetite is controlled by a key molecule called leptin. Leptin is a hormone which is produced by our fat cells, and is delivered by the blood to the brain to signal the brain that we are full and can stop eating,” explains Dr. Maia Kokoeva who is affiliated both with McGill University and the Research Institute of the McGill University Health Centre. “But even though receptors for leptin were discovered soon after in the hypothalamus, a brain area that regulates food intake and body weight, it has remained unclear how exactly leptin is detected.”
So about four years ago, Kokoeva and her team set out to explore which brain cells might play a role in the process of leptin sensing and weight gain. The answer, it turns out, lies in the median eminence.
“Protection” and “preservation” cells in a busy place
The median eminence is a brain structure at the base of the hypothalamus. It is a bit like a busy hub or market place through which hormones and molecules of various kinds travel in both directions between the brain and the bloodstream to ensure that the body functions smoothly.
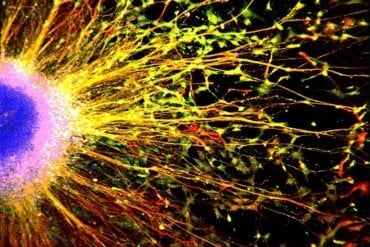
The McGill research team has now discovered that without a particular group of cells (known as NG2-glia cells) in place in the median eminence, the leptin receptors in the brain never receive the messages from the body telling it that it is sated.
“Most of the brain is a well-protected fortress, designed to shelter delicate nerve cells,” says Kokoeva. “The median eminence is outside these protections, and so can be a dangerous environment for the nerve cells that detect leptin. We think that the NG2-glia cells act to support and shelter the leptin receptor neurons, enabling them to instruct the body when to stop eating.”
Crucial role of the median eminence in weight gain
“We developed an interest in NG2-glia cells in this specific part of the brain because unlike neurons, during much of our adult lives these cells are constantly dividing and they do so most actively in the median eminence,” says Tina Djogo, a McGill doctoral student and one of two lead authors on the study which was published this week in Cell Metabolism. “But though these cells were first described about thirty years ago it has been difficult so far to pinpoint their exact functions in the adult brain.”
Because of their particularly high turnover in the median eminence, the researchers wondered if the NG2-glia cells might play a role in leptin sensing and therefore in appetite control. So they used a drug to kill the NG2-glia cells in the median eminence of a group of mice and then watched to see whether there was a difference in food intake. The results were stunning.
Within three days after they started to receive the medication, some of the mice dubbed “gainers” had already started to eat more compared with the control group of mice who had not received medication. And by 30 days afterwards, the weight of some of the mice had doubled – from 25 grams to around 50 grams.
“But what was most exciting to us, was that even though NG2-glia are found across the brain” explains Sarah Robins, a research associate who was also a lead author on this study, “it was only when we removed these cells from the median eminence that we saw this clear increase in body weight.”
A possible explanation for weight gain in brain tumour patients
The researchers then corroborated the role of the NG2-glia cells in the median eminence in appetite control through experiments using genetically modified mice, and also by using irradiation. This latter discovery suggested an explanation for a previously unexplained phenomenon in human brain cancer survivors.

“People who have been treated for brain tumours using radiation to block cell proliferation often become overweight,” says Kokoeva. “However, there has never been any satisfactory explanation, but our experiments in mice now suggests that the reason for this weight gain may be the loss of NG2-glia in the median eminence as a result of radiation.”
The researchers are hopeful that the identification of NG2-glia in the median eminence as crucial elements in body weight and appetite control will pave the way to new targeted anti-obesity approaches directed towards maintaining or raising the NG2-glia population in the median eminence.
Funding: This research was supported by Canadian Institutes of Health Research operating grants, the Deutsche Forschungsgemeinschaft, the Bundesministerium fur Bildung und Forschung (BMBF), the Excellence Initiative Synergy, and the Friedrich Bauer Stiftung.
Source: McGill University
Image Source: The image is credited to Matthew R. Brier.
Original Research: Abstract for “Adult NG2-Glia Are Required for Median Eminence-Mediated Leptin Sensing and Body Weight Control” by Tina Djogo, Sarah C. Robins, Sarah Schneider, Darya Kryzskaya, Xiaohong Liu, Andrew Mingay, Colleen J. Gillon, Joo Hyun Kim, Kai-Florian Storch, Ulrich Boehm, Charles W. Bourque, Thomas Stroh, Leda Dimou, and Maia V. Kokoeva in Cell Metabolism. Published online May 10 2016 doi:10.1016/j.cmet.2016.04.013
Abstract
Adult NG2-Glia Are Required for Median Eminence-Mediated Leptin Sensing and Body Weight Control
Highlights
•Pharmacological and genetic ablation of NG2-glia, but not microglia, leads to obesity
•NG2-glial ablation causes LepR processes in the median eminence to degenerate
•Arcuate nucleus LepR neurons lose responsiveness to leptin after NG2-glia ablation
•X-irradiation aimed at the median eminence is sufficient for weight gain induction
Summary
While leptin is a well-known regulator of body fat mass, it remains unclear how circulating leptin is sensed centrally to maintain energy homeostasis. Here we show that genetic and pharmacological ablation of adult NG2-glia (also known as oligodendrocyte precursors), but not microglia, leads to primary leptin resistance and obesity in mice. We reveal that NG2-glia contact the dendritic processes of arcuate nucleus leptin receptor (LepR) neurons in the median eminence (ME) and that these processes degenerate upon NG2-glia elimination, which explains the consequential attenuation of these neurons’ molecular and electrical responses to leptin. Our data therefore indicate that LepR dendrites in the ME represent the principal conduits of leptin’s anorexigenic action and that NG2-glia are essential for their maintenance. Given that ME-directed X-irradiation confirmed the pharmacological and genetically mediated ablation effects on body weight, our findings provide a rationale for the known obesity risk associated with cranial radiation therapy.
“Adult NG2-Glia Are Required for Median Eminence-Mediated Leptin Sensing and Body Weight Control” by Tina Djogo, Sarah C. Robins, Sarah Schneider, Darya Kryzskaya, Xiaohong Liu, Andrew Mingay, Colleen J. Gillon, Joo Hyun Kim, Kai-Florian Storch, Ulrich Boehm, Charles W. Bourque, Thomas Stroh, Leda Dimou, and Maia V. Kokoeva in Cell Metabolism. Published online May 10 2016 doi:10.1016/j.cmet.2016.04.013