Summary: Researchers describe how to prepare a specific type of aggregate of the amyloid-beta protein with the ability to perforate the cell membrane.
Source: IRB Barcelona.
Researchers at IRB Barcelona describe for the first time how to prepare a specific type of aggregate of the amyloid-beta protein with the ability to perforate the cell membrane.
The brains of millions of people suffering from Alzheimer’s disease are slowly and inescapably being depleted of neurons. However, the cause of neuronal death is still unknown. Several studies propose that the interaction of the amyloid-beta protein with the neuronal membrane causes neurotoxicity.
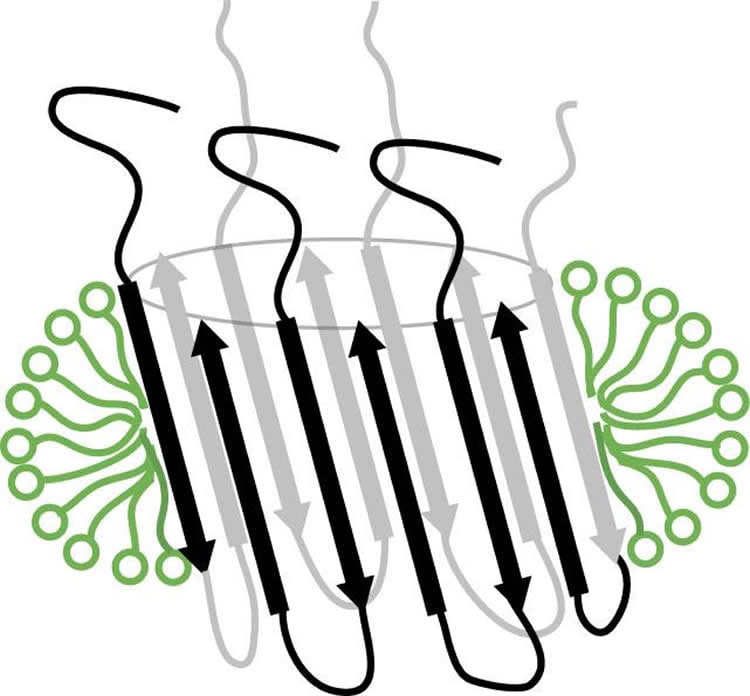
Today, a study headed by associate researcher Natàlia Carulla, in the Peptides and Proteins Laboratory at the Institute for Research in Biomedicine (IRB Barcelona), in collaboration with scientists at the University of Leuven (Belgium), the University of Groningen (the Netherlands), and the Technical-Scientific Services of the University of Barcelona, describes for the first time a strategy through which to obtain a membrane-associated amyloid-beta aggregate that adopts a specific barrel-like structure. This type of structure -which is present in other proteins found in nature- has the capacity to form pores in cell membranes. Interpreted in the context of Alzheimer’s disease, this discovery suggests that the aggregate can also perforate the membrane of neurons, alter the equilibrium of these cells, and trigger their death.
The description of the barrel-shaped aggregates in the journal Proceedings of the National Academy of Sciences (PNAS) is the first step to establish the involvement of this aggregate in Alzheimer’s disease. Confirming the involvement would validate it as a new therapeutic target for this condition. The scientists made this discovery in the lab by studying amyloid-beta protein in simple membrane model systems.
Attack to the amyloid-beta protein
Dr. Natàlia Carulla develops methods to identify the nature and structure of the toxic amyloid-beta species. “Everyone talks about the relevance of oligomers (aggregates made of the same protein unit) of amyloid-beta and their contribution to Alzheimer’s disease, but if we don’t know which structural features characterize them, we won’t know what to look for, and therefore we won’t be able to demonstrate or disprove anything,” explains Carulla. “We don’t know whether this species is neurotoxic, but now that we know that it adopts a specific barrel-like structure, we can study it in the context of the disease since we know what to look for,” say Montserrat Serra-Batiste and Martí Ninot-Pedrosa from IRB Barcelona, first and second authors of the paper, respectively.
The next steps involve obtaining the 3D structure of the aggregate and obtaining specific antibodies that recognize it. Afterwards, the researchers will study the function of this aggregate in cultured neurons derived from patients with Alzheimer’s disease. These will be decisive experiments for the validation of this specific amyloid-beta aggregate as a therapeutic target.
Funding: Natàlia Carulla’s research is supported by funds from “La Marató de TV3” awarded in 2014, and the “Plan Nacional”and ERDFs from the Ministry of Economy and Competitiveness.
Source: Sonia Armengou – IRB Barcelona
Image Source: NeuroscienceNews.com image is credited to Natàlia Carulla, IRB Barcelona.
Original Research: Abstract for “Aβ42 assembles into specific β-barrel pore-forming oligomers in membrane-mimicking environments” by Montserrat Serra-Batiste, Martí Ninot-Pedrosa, Mariam Bayoumi, Margarida Gairí, Giovanni Maglia, and Natàlia Carulla in PNAS. Published online August 2 2016 doi:10.1073/pnas.1605104113
[cbtabs][cbtab title=”MLA”]IRB Barcelona. “Obtaining an Amyloid Beta Aggregate That May Underlie Neuronal Death in Alzheimer’s.” NeuroscienceNews. NeuroscienceNews, 13 September 2016.
<https://neurosciencenews.com/alzheimers-amyloid-aggregate-5039/>.[/cbtab][cbtab title=”APA”]IRB Barcelona. (2016, September 13). Obtaining an Amyloid Beta Aggregate That May Underlie Neuronal Death in Alzheimer’s. NeuroscienceNews. Retrieved September 13, 2016 from https://neurosciencenews.com/alzheimers-amyloid-aggregate-5039/[/cbtab][cbtab title=”Chicago”]IRB Barcelona. “Obtaining an Amyloid Beta Aggregate That May Underlie Neuronal Death in Alzheimer’s.” https://neurosciencenews.com/alzheimers-amyloid-aggregate-5039/ (accessed September 13, 2016).[/cbtab][/cbtabs]
Abstract
Aβ42 assembles into specific β-barrel pore-forming oligomers in membrane-mimicking environments
The formation of amyloid-β peptide (Aβ) oligomers at the cellular membrane is considered to be a crucial process underlying neurotoxicity in Alzheimer’s disease (AD). Therefore, it is critical to characterize the oligomers that form within a membrane environment. To contribute to this characterization, we have applied strategies widely used to examine the structure of membrane proteins to study the two major Aβ variants, Aβ40 and Aβ42. Accordingly, various types of detergent micelles were extensively screened to identify one that preserved the properties of Aβ in lipid environments—namely the formation of oligomers that function as pores. Remarkably, under the optimized detergent micelle conditions, Aβ40 and Aβ42 showed different behavior. Aβ40 aggregated into amyloid fibrils, whereas Aβ42 assembled into oligomers that inserted into lipid bilayers as well-defined pores and adopted a specific structure with characteristics of a β-barrel arrangement that we named β-barrel pore-forming Aβ42 oligomers (βPFOsAβ42). Because Aβ42, relative to Aβ40, has a more prominent role in AD, the higher propensity of Aβ42 to form βPFOs constitutes an indication of their relevance in AD. Moreover, because βPFOsAβ42 adopt a specific structure, this property offers an unprecedented opportunity for testing a hypothesis regarding the involvement of βPFOs and, more generally, membrane-associated Aβ oligomers in AD.
“Aβ42 assembles into specific β-barrel pore-forming oligomers in membrane-mimicking environments” by Montserrat Serra-Batiste, Martí Ninot-Pedrosa, Mariam Bayoumi, Margarida Gairí, Giovanni Maglia, and Natàlia Carulla in PNAS. Published online August 2 2016 doi:10.1073/pnas.1605104113






