Summary: Researchers have developed a new machine learning algorithm that can make accurate predictions about cognitive decline leading to Alzheimer’s. The algorithm, researchers say, may help doctors stream people onto a preventative path and offer treatment sooner.
Source: McGill University.
A team of scientists has successfully trained a new artificial intelligence (AI) algorithm to make accurate predictions regarding cognitive decline leading to Alzheimer’s disease.
Dr. Mallar Chakravarty, a computational neuroscientist at the Douglas Mental Health University Institute, and his colleagues from the University of Toronto and the Centre for Addiction and Mental Health, designed an algorithm that learns signatures from magnetic resonance imaging (MRI), genetics, and clinical data. This specific algorithm can help predict whether an individual’s cognitive faculties are likely to deteriorate towards Alzheimer’s in the next five years.
“At the moment, there are limited ways to treat Alzheimer’s and the best evidence we have is for prevention. Our AI methodology could have significant implications as a ‘doctor’s assistant’ that would help stream people onto the right pathway for treatment. For example, one could even initiate lifestyle changes that may delay the beginning stages of Alzheimer’s or even prevent it altogether,” says Chakravarty, an Assistant Professor in McGill University’s Department of Psychiatry.
The findings, published in PLOS Computational Biology, used data from the Alzheimer’s Disease NeuroImaging Initiative. The researchers trained their algorithms using data from more than 800 people ranging from normal healthy seniors to those experiencing mild cognitive impairment, and Alzheimer’s disease patients. They replicated their results within the study on an independently collected sample from the Australian Imaging and Biomarkers Lifestyle Study of Ageing.
Can the predictions be improved with more data?
“We are currently working on testing the accuracy of predictions using new data. It will help us to refine predictions and determine if we can predict even farther into the future,” says Chakravarty. With more data, the scientists would be able to better identify those in the population at greatest risk for cognitive decline leading to Alzheimer’s.
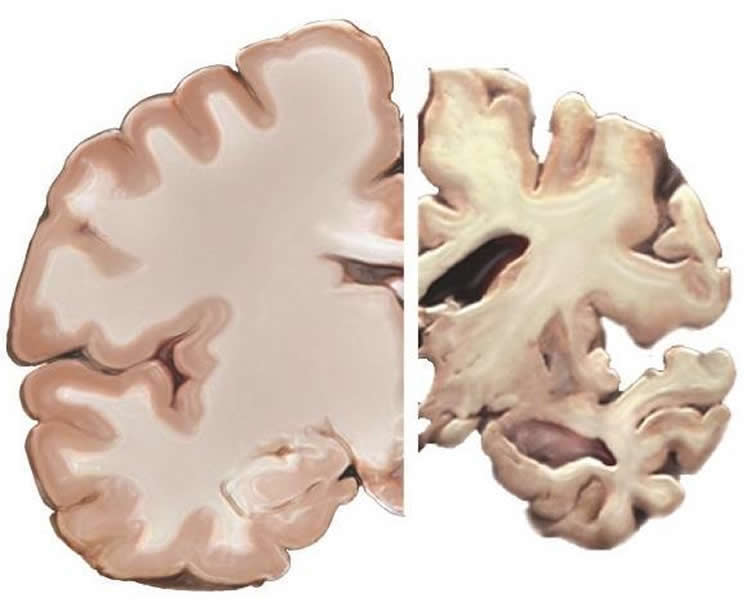
According to the Alzheimer Society of Canada, 564,000 Canadians had Alzheimer’s or another form of dementia in 2016. The figure will rise to 937,000 within 15 years.
Worldwide, around 50million people have dementia and the total number is projected to reach 82million in 2030 and 152 in 2050, according to the World Health Organization. Alzheimer’s disease, the most common form of dementia, may contribute to 60-70% of cases. Presently, there is no truly effective treatment for this disease.
Funding: This work was funded by the Canadian Institutes of Health Research, the Natural Sciences andEngineering Research Council of Canada, the Fonds de recherche du Québec–Santé, Weston Brain Institute, Michael J. Fox Foundation for Parkinson’s Research, Alzheimer’s Society, Brain Canada, and the McGill University Healthy Brains for Healthy Lives – Canada First Research Excellence Fund.
Source: Cynthia Lee – McGill University
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is in the public domain.
Original Research: Open access research for “Modeling and prediction of clinical symptom trajectories in Alzheimer’s disease using longitudinal data” by Nikhil Bhagwat, Joseph D. Viviano, Aristotle N. Voineskos, M. Mallar Chakravarty, Alzheimer’s Disease Neuroimaging Initiative in PLOS Computational Biology. Published September 14 2018.
doi:10.1371/journal.pcbi.1006376
[cbtabs][cbtab title=”MLA”]McGIll University”AI Could Predict Cognitive Decline Leading to Alzheimer’s.” NeuroscienceNews. NeuroscienceNews, 4 October 2018.
<https://neurosciencenews.com/ai-alzheimers-cognition-9960/>.[/cbtab][cbtab title=”APA”]McGIll University(2018, October 4). AI Could Predict Cognitive Decline Leading to Alzheimer’s. NeuroscienceNews. Retrieved October 4, 2018 from https://neurosciencenews.com/ai-alzheimers-cognition-9960/[/cbtab][cbtab title=”Chicago”]McGIll University”AI Could Predict Cognitive Decline Leading to Alzheimer’s.” https://neurosciencenews.com/ai-alzheimers-cognition-9960/ (accessed October 4, 2018).[/cbtab][/cbtabs]
Abstract
Modeling and prediction of clinical symptom trajectories in Alzheimer’s disease using longitudinal data
Computational models predicting symptomatic progression at the individual level can be highly beneficial for early intervention and treatment planning for Alzheimer’s disease (AD). Individual prognosis is complicated by many factors including the definition of the prediction objective itself. In this work, we present a computational framework comprising machine-learning techniques for 1) modeling symptom trajectories and 2) prediction of symptom trajectories using multimodal and longitudinal data. We perform primary analyses on three cohorts from Alzheimer’s Disease Neuroimaging Initiative (ADNI), and a replication analysis using subjects from Australian Imaging, Biomarker & Lifestyle Flagship Study of Ageing (AIBL). We model the prototypical symptom trajectory classes using clinical assessment scores from mini-mental state exam (MMSE) and Alzheimer’s Disease Assessment Scale (ADAS-13) at nine timepoints spanned over six years based on a hierarchical clustering approach. Subsequently we predict these trajectory classes for a given subject using magnetic resonance (MR) imaging, genetic, and clinical variables from two timepoints (baseline + follow-up). For prediction, we present a longitudinal Siamese neural-network (LSN) with novel architectural modules for combining multimodal data from two timepoints. The trajectory modeling yields two (stable and decline) and three (stable, slow-decline, fast-decline) trajectory classes for MMSE and ADAS-13 assessments, respectively. For the predictive tasks, LSN offers highly accurate performance with 0.900 accuracy and 0.968 AUC for binary MMSE task and 0.760 accuracy for 3-way ADAS-13 task on ADNI datasets, as well as, 0.724 accuracy and 0.883 AUC for binary MMSE task on replication AIBL dataset.






