Summary: The function of the opioid system is connected to eating triggered by external stimuli. The findings could lead to the development of new therapies that target the opioid system to reduce obesity.
Source: University of Turku
Brain regulation of feeding behavior traits has remained incompletely understood. In their latest study, researchers at the Turku PET Centre, Finland, discovered a connection between the function of the opioid system and food craving triggered by appetitive external stimuli.
Animal studies have established that the brain’s opioid and endocannabinoid systems are important in regulating eating behavior and mediate the food reward experience. For instance, alterations in these systems’ signaling have been associated with obesity. In general, both internal signals of the body, such as fluctuation in blood sugar levels, and external stimuli, such as food advertisements, can spark an appetite in humans.
In their new study, researchers at the University of Turku, Finland, investigated the connection between the brain’s opioid and endocannabinoid signaling and different types of eating behavior. They discovered that the function of the opioid system is connected to eating triggered by external stimuli.
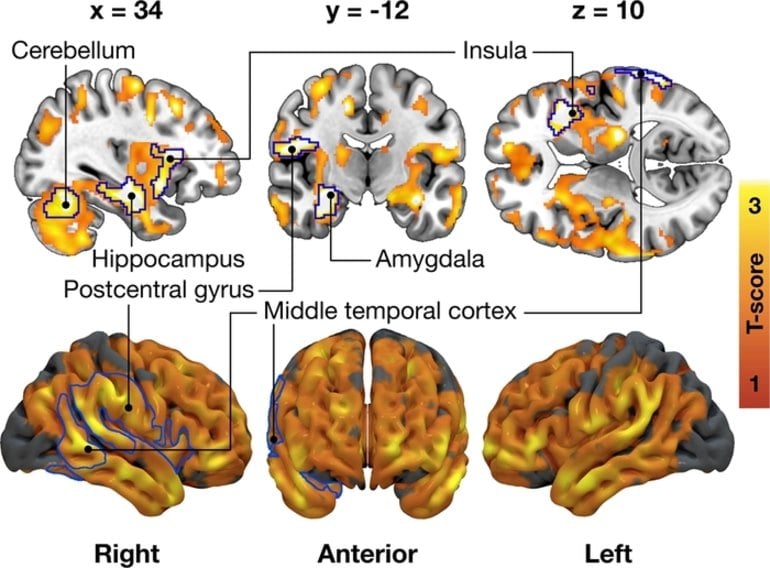
“The less binding sites there were for the opioids, the greater was the tendency to eat in response to external stimuli, such as seeing appetizing food. Moreover, the number of binding sites for endocannabinoids was connected to several different types of eating behavior, describes first author,” Doctoral Candidate Tatu Kantonen from the University of Turku.
According to Kantonen, the results indicate that especially the opioid system could be a potential target for anti-obesity drugs in humans.
The research data was obtained from the AIVO database hosted by the Turku PET Centre.
About this neuroscience research news
Author: Tuomas Koivula
Source: University of Turku
Contact: Tuomas Koivula – University of Turku
Image: The image is credited to University of Turku
Original Research: Open access.
“Cerebral μ-opioid and CB1 receptor systems have distinct roles in human feeding behavior” by Tatu Kantonen, Tomi Karjalainen, Laura Pekkarinen, Janne Isojärvi, Kari Kalliokoski, Valtteri Kaasinen, Jussi Hirvonen, Pirjo Nuutila & Lauri Nummenmaa. Translational Psychiatry
Abstract
Cerebral μ-opioid and CB1 receptor systems have distinct roles in human feeding behavior
Eating behavior varies greatly between individuals, but the neurobiological basis of these trait-like differences in feeding remains poorly understood.
Central μ-opioid receptors (MOR) and cannabinoid CB1 receptors (CB1R) regulate energy balance via multiple neural pathways, promoting food intake and reward. Because obesity and eating disorders have been associated with alterations in the brain’s opioid and endocannabinoid signaling, the variation in MOR and CB1R system function could potentially underlie distinct eating behavior phenotypes.
In this retrospective positron emission tomography (PET) study, we analyzed [11C]carfentanil PET scans of MORs from 92 healthy subjects (70 males and 22 females), and [18F]FMPEP-d2 scans of CB1Rs from 35 subjects (all males, all also included in the [11C]carfentanil sample).
Eating styles were measured with the Dutch Eating Behavior Questionnaire (DEBQ). We found that lower cerebral MOR availability was associated with increased external eating—individuals with low MORs reported being more likely to eat in response to environment’s palatable food cues. CB1R availability was associated with multiple eating behavior traits.
We conclude that although MORs and CB1Rs overlap anatomically in brain regions regulating food reward, they have distinct roles in mediating individual feeding patterns. Central MOR system might provide a pharmacological target for reducing individual’s excessive cue-reactive eating behavior.






