Summary: Researchers discovered decreased function in a protein critical for synaptic function in those with Huntington’s disease.
Source: KIST
Huntington’s disease (HD) is a hereditary brain disease caused by a mutation in the huntingtin gene.
HD is a neurodegenerative disease without a cure that, after the onset of the disease at around 40 years of age, causes changes in personality and symptoms of dementia along with uncontrollable convulsive movements, ultimately leading to death. It is known that such HD symptoms are caused by the destruction of brain cells in the striatum due to problems occurring in synapses that are crucial to brain function during the progression of the disease.
However, the specific mechanism behind brain dysfunction during the progression of HD has not been fully elucidated.
The research team lead by Dr. Jihye Seong and Dr. Hoon Ryu, principal researchers at the Brain Science Institute (BSI) of Korea Institute of Science and Technology (KIST, President Seokjin Yoon), was said to have found significantly reduced activity of focal adhesion kinase (FAK) proteins that play an important role in neurite motility and proper synapse formation in the brain tissues of patients with HD.
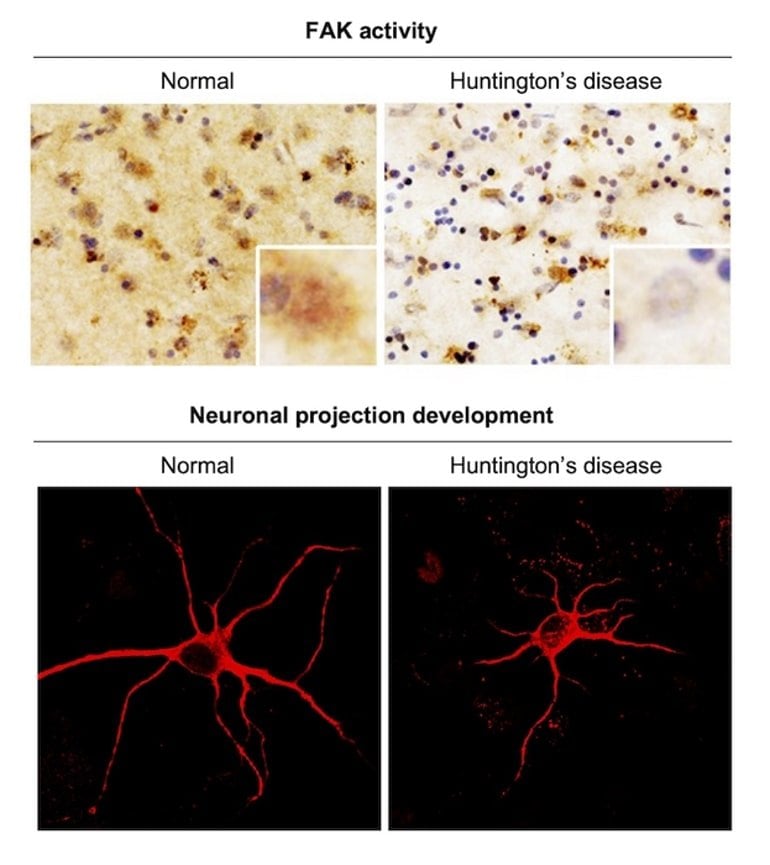
Activated FAK proteins play an important role in brain function as they are essential in neurite motility and proper synapse formation. The KIST research team identified a significant reduction in FAK activity in HD cells and mouse models, as well as brain tissues of HD patients. These results were also verified through accurate measurements of FAK activity in live cells using a fluorescence resonance energy transfer (FRET)-based biosensor.
Phosphatidylinositol 4,5-biphosphate (PIP2), a phospholipid found in the cell membrane, is essential for the activation of FAK proteins.
Using super-resolution structured illumination microscopy, the research team found that PIP2 in HD cells was unusually strongly bound to the mutant huntingtin protein, inhibiting proper distribution of PIP2 throughout the cell membrane. This abnormal distribution of PIP2 inhibits FAK activation, which hinders proper synaptic function, causing brain dysfunction in the early stages of HD.
Dr. Seong said, “The pathological mechanisms of synaptic dysfunction in patients with Huntington’s disease revealed through this study could be utilized as a therapeutic target for the treatment of brain dysfunction.” Dr. Ryu said, “Because the results of this study show the pathological mechanisms found in actual brain tissues of patients with HD, I believe it has a greater significance in suggesting a new therapeutic target for human degenerative brain diseases.”
About this Huntington’s disease research news
Author: Mira Lee
Source: KIST
Contact: Mira Lee – KIST
Image: The image is credited to KIST
Original Research: Closed access.
“Decreased FAK activity and focal adhesion dynamics impair proper neurite formation of medium spiny neurons in Huntington’s disease” by Seong, Jihye et al. Acta Neuropathologica
Abstract
Decreased FAK activity and focal adhesion dynamics impair proper neurite formation of medium spiny neurons in Huntington’s disease
Huntington’s disease (HD) is a neurodegenerative disorder caused by a polyglutamine expansion in the protein huntingtin (HTT) [55]. While the final pathological consequence of HD is the neuronal cell death in the striatum region of the brain, it is still unclear how mutant HTT (mHTT) causes synaptic dysfunctions at the early stage and during the progression of HD.
Here, we discovered that the basal activity of focal adhesion kinase (FAK) is severely reduced in a striatal HD cell line, a mouse model of HD, and the human post-mortem brains of HD patients.
In addition, we observed with a FRET-based FAK biosensor [59] that neurotransmitter-induced FAK activation is decreased in HD striatal neurons. Total internal reflection fluorescence (TIRF) imaging revealed that the reduced FAK activity causes the impairment of focal adhesion (FA) dynamics, which further leads to the defect in filopodial dynamics causing the abnormally increased number of immature neurites in HD striatal neurons.
Therefore, our results suggest that the decreased FAK and FA dynamics in HD impair the proper formation of neurites, which is crucial for normal synaptic functions [52].
We further investigated the molecular mechanism of FAK inhibition in HD and surprisingly discovered that mHTT strongly associates with phosphatidylinositol 4,5-biphosphate, altering its normal distribution at the plasma membrane, which is crucial for FAK activation [14, 60].
Therefore, our results provide a novel molecular mechanism of FAK inhibition in HD along with its pathological mechanism for synaptic dysfunctions during the progression of HD.






