Summary: AgRP neurons in the hypothalamus play a critical role in shaping the structure and function of the prefrontal cortex in mice. The findings shed light on how the prefrontal cortex is altered in disorders such as schizophrenia.
Source: Yale
The prefrontal cortex region of the human brain is responsible for a range of complex functions from decision-making to certain types of memory.
When something goes wrong in this part of the brain, it can be very detrimental for cognition and behavior. In fact, dysfunction in the prefrontal cortex is linked to several psychiatric illnesses, including schizophrenia and major depressive disorder.
Yale researchers and their colleagues in Hungary have discovered that cells in the hypothalamus—an area of the brain that controls functions like hunger and body temperature—play a major role in shaping the structure and function of the prefrontal cortex in mice, a finding that could offer clues into how this region of the brain is altered in disease and open new avenues for treatment.
They reported their findings July 29 in the journal Molecular Psychiatry.
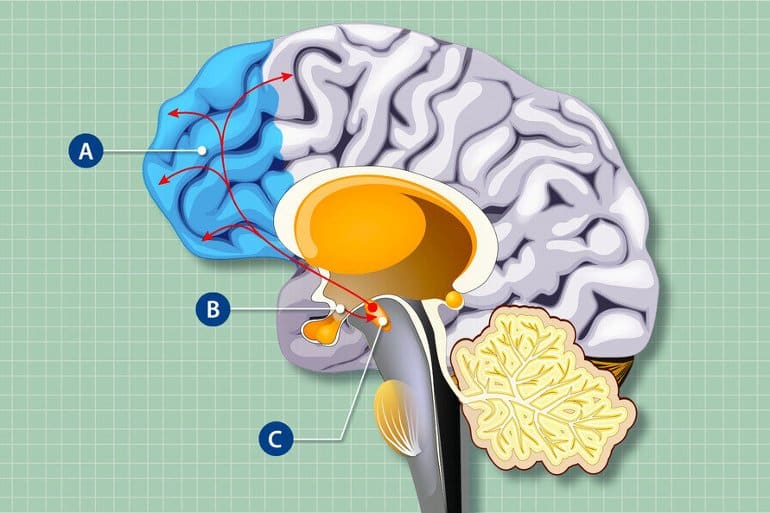
For the study, the researchers focused on agouti-related peptide (AgRP) neurons located in the hypothalamus region of the brain. These neurons control hunger and regulate both feeding behaviors and non-feeding behaviors like reward-seeking and offspring-parent bonding, among others.
When the researchers impaired AgRP neurons in mice, they found there were fewer neurons in the prefrontal cortex than there were in healthy animals.
“The neurons that remained were smaller than usual and behaved differently in response to cues from the body and signals from surrounding cells,” said Tamas Horvath, the Jean and David W. Wallace Professor of Comparative Medicine at Yale and senior author of the study.
AgRP neurons do not have strong direct connections to the cortex. But they do project to other brain regions that connect to the prefrontal cortex.
Horvath and his colleagues found that the neurons in one of these areas—a midbrain region known as the ventral tegmental area—were overactive when AgRP neurons were impaired. Those overactive neurons then released more dopamine, a neurotransmitter, in the prefrontal cortex than is typical in healthy mice, which, in turn, negatively affected mouse behavior.
For instance, they found that the mice moved around much more and had abnormal startle responses.
It makes sense that these neurons that control hunger and feeding would affect the cortex and behavior, says Horvath.
“When you are hungry, you need all of your behaviors lined up so you can find food and eat it,” he said. “And when you are no longer hungry, you have to change your behaviors to focus on what’s important in that moment.”
After uncovering the effects impaired AgRP cells had on the cortex, the researchers then tried to avert them. They found that clozapine—a type of antipsychotic that blocks dopamine from acting and is used to treat schizophrenia—was able to prevent some of these problems, including neuron loss, when given at the right time.
When it comes to these cortical changes, Horvath said, the timing is crucial. In the study, impairments began to emerge during puberty when the brain is still developing and vulnerable. That’s also when clozapine administration had an impact.
“And this tells us that if you play with homeostatic functions in that specific time period, say by dieting or overeating, you can have long-lasting effects on your cortical functions,” he said.
The importance of this period of development may shed light on psychiatric illness, which can often emerge in late adolescence, and why substance use during that time can have long-lasting effects on behavior, physiology, and disease.
These findings may also provide a new target for treatment. AgRP cells in the hypothalamus are outside of the blood-brain barrier, a feature of the brain that protects it from harmful substances and prevents many medications from reaching brain tissue.
“This means these cells are readily available for intervention,” said Horvath. “Perhaps they can be exploited to alter disorders of higher brain regions.”
The study also adds to a growing body of evidence that the function of the cortex is influenced by more primitive regions of the brain and by other parts of the body. Horvath recently showed how AgRP cells can also affect the cortex via mediation of the liver.
“The fundamental message here is that in the brain, there is very diffuse communication through different pathways,” said Horvath.
“Primitive areas like the hypothalamus influence higher cortical regions through innumerable number of communications, which includes brain processes, but also peripheral tissues.
“We are back to the debate between Camillo Golgi and Ramón y Cajal, who shared the Nobel Prize in 1906, but disagreed on the principles of how the brain works. Our results seem to favor forgotten arguments of Camillo Golgi.”
About this neuroscience research news
Author: Mallory Locklear
Source: Yale
Contact: Mallory Locklear – Yale
Image: The image is credited to Yale
Original Research: Open access.
“AgRP neurons control structure and function of the medial prefrontal cortex” by Bernardo Stutz et al. Molecular Psychiatry
Abstract
AgRP neurons control structure and function of the medial prefrontal cortex
Hypothalamic agouti-related peptide and neuropeptide Y-expressing (AgRP) neurons have a critical role in both feeding and non-feeding behaviors of newborn, adolescent, and adult mice, suggesting their broad modulatory impact on brain functions.
Here we show that constitutive impairment of AgRP neurons or their peripubertal chemogenetic inhibition resulted in both a numerical and functional reduction of neurons in the medial prefrontal cortex (mPFC) of mice.
These changes were accompanied by alteration of oscillatory network activity in mPFC, impaired sensorimotor gating, and altered ambulatory behavior that could be reversed by the administration of clozapine, a non-selective dopamine receptor antagonist. The observed AgRP effects are transduced to mPFC in part via dopaminergic neurons in the ventral tegmental area and may also be conveyed by medial thalamic neurons.
Our results unmasked a previously unsuspected role for hypothalamic AgRP neurons in control of neuronal pathways that regulate higher-order brain functions during development and in adulthood.







