Summary: Researchers have demonstrated the role of lysosomal vesicles in transporting α-synuclein aggregates.
Source: Institut Pasteur.
Scientists from the Institut Pasteur have demonstrated the role of lysosomal vesicles in transporting α-synuclein aggregates, responsible for Parkinson’s and other neurodegenerative diseases, between neurons. These proteins move from one neuron to the next in lysosomal vesicles which travel along the “tunneling nanotubes” between cells. These findings were published in The EMBO Journal.
Synucleinopathies, a group of neurodegenerative diseases including Parkinson’s disease, are characterized by the pathological deposition of aggregates of the misfolded α-synuclein protein into inclusions throughout the central and peripheral nervous system. Intercellular propagation (from one neuron to the next) of α-synuclein aggregates contributes to the progression of the neuropathology, but little was known about the mechanism by which spread occurs.
In this study, scientists from the Membrane Traffic and Pathogenesis Unit, directed by Chiara Zurzolo at the Institut Pasteur, used fluorescence microscopy to demonstrate that pathogenic α-synuclein fibrils travel between neurons in culture, inside lysosomal vesicles through tunneling nanotubes (TNTs), a new mechanism of intercellular communication.
After being transferred via TNTs, α-synuclein fibrils are able to recruit and induce aggregation of the soluble α-synuclein protein in the cytosol of cells receiving the fibrils, thus explaining the propagation of the disease. The scientists propose that cells overloaded with α-synuclein aggregates in lysosomes dispose of this material by hijacking TNT-mediated intercellular trafficking. However, this results in the disease being spread to naive neurons.
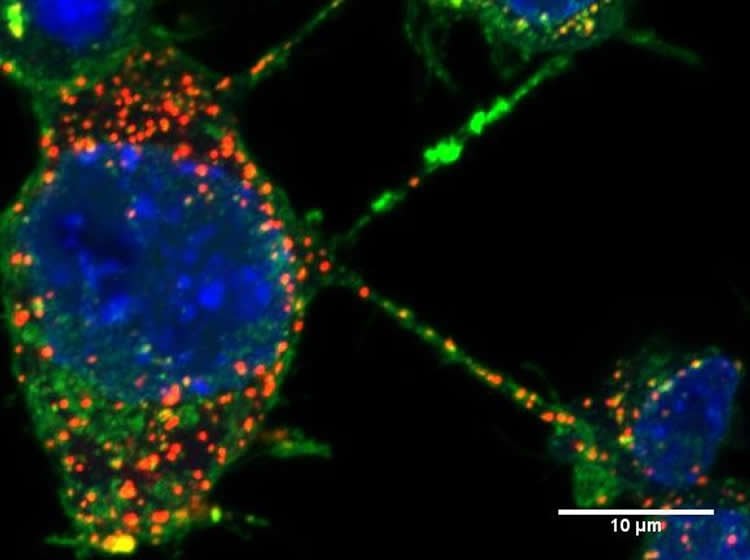
This study demonstrates that TNTs play a significant part in the intercellular transfer of α-synuclein fibrils and reveals the specific role of lysosomes in this process. This represents a major breakthrough in understanding the mechanisms underlying the progression of synucleinopathies.
These compelling findings, together with previous reports from the same team, point to the general role of TNTs in the propagation of prion-like proteins in neurodegenerative diseases and identify TNTs as a new therapeutic target to combat the progression of these incurable diseases.
Source: Myriam Rebeyrotte – Institut Pasteur
Image Source: This NeuroscienceNews.com image is credited to Institut Pasteur.
Original Research: Abstract for “Tunneling nanotubes spread fibrillar α‐synuclein by intercellular trafficking of lysosomes” by Saïda Abounit, Luc Bousset, Frida Loria, Seng Zhu, Fabrice de Chaumont, Laura Pieri, Jean‐Christophe Olivo‐Marin, Ronald Melki, and Chiara Zurzolo in EMBO Journal. Published online July 22 2016 doi:10.15252/embj.201593411
[cbtabs][cbtab title=”MLA”]Institut Pasteur. “Tunneling Nanotubes Between Neurons Enable the Spread of Parkinson’s.” NeuroscienceNews. NeuroscienceNews, 22 August 2016.
<https://neurosciencenews.com/parkinsons-nanotubes-lysosomes-4885/>.[/cbtab][cbtab title=”APA”]Institut Pasteur. (2016, August 22). Tunneling Nanotubes Between Neurons Enable the Spread of Parkinson’s. NeuroscienceNew. Retrieved August 22, 2016 from https://neurosciencenews.com/parkinsons-nanotubes-lysosomes-4885/[/cbtab][cbtab title=”Chicago”]Institut Pasteur. “Tunneling Nanotubes Between Neurons Enable the Spread of Parkinson’s.” https://neurosciencenews.com/parkinsons-nanotubes-lysosomes-4885/ (accessed August 22, 2016).[/cbtab][/cbtabs]
Abstract
Tunneling nanotubes spread fibrillar α‐synuclein by intercellular trafficking of lysosomes
Synucleinopathies such as Parkinson’s disease are characterized by the pathological deposition of misfolded α‐synuclein aggregates into inclusions throughout the central and peripheral nervous system. Mounting evidence suggests that intercellular propagation of α‐synuclein aggregates may contribute to the neuropathology; however, the mechanism by which spread occurs is not fully understood. By using quantitative fluorescence microscopy with co‐cultured neurons, here we show that α‐synuclein fibrils efficiently transfer from donor to acceptor cells through tunneling nanotubes (TNTs) inside lysosomal vesicles. Following transfer through TNTs, α‐synuclein fibrils are able to seed soluble α‐synuclein aggregation in the cytosol of acceptor cells. We propose that donor cells overloaded with α‐synuclein aggregates in lysosomes dispose of this material by hijacking TNT‐mediated intercellular trafficking. Our findings thus reveal a possible novel role of TNTs and lysosomes in the progression of synucleinopathies.
“Tunneling nanotubes spread fibrillar α‐synuclein by intercellular trafficking of lysosomes” by Saïda Abounit, Luc Bousset, Frida Loria, Seng Zhu, Fabrice de Chaumont, Laura Pieri, Jean‐Christophe Olivo‐Marin, Ronald Melki, and Chiara Zurzolo in EMBO Journal. Published online July 22 2016 doi:10.15252/embj.201593411







