Summary: The direct contact of mGluR4 receptors with other key proteins plays a significant role in the regulation of synaptic activity.
Source: University of Wurzburg
When people think of glutamate, the first thing they remember is the flavor enhancer that is often used in Asian cuisine. Glutamate is also an important messenger substance in the nervous system of humans. There it plays a role in learning processes and memory. Some Alzheimer drugs, for example, slow down the progression of the disease by inhibiting the effect of glutamate.
In the nervous system, glutamate acts as a signal transmitter at the synapses. There, it binds to specific receptors of which there are several types. The metabotropic glutamate receptor of type 4 (mGluR4) plays a decisive role in this system.
Direct contact to other proteins
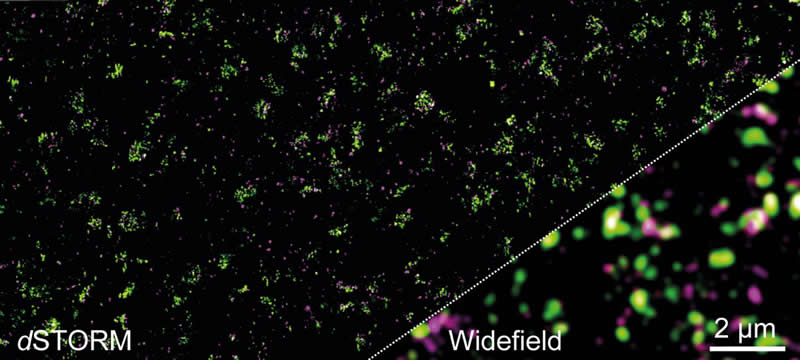
Until now, not much was known about the distribution of this receptor in the active zones of synapses. It is now clear that the majority of mGluR4 receptors are located in groups of one to two units on average in the presynaptic membrane. There they are often in direct contact with calcium channels and the protein Munc-18-1, which is important for the release of messengers.
This is reported in the journal Science Advances by a research team led by Professor Markus Sauer from the Biocenter of Julius-Maximilians-Universität (JMU) Würzburg in Bavaria, Germany, and Professor Davide Calebiro from the University of Birmingham in England. “Our data indicate that the direct contact of mGluR4 receptors with other key proteins plays a major role in the regulation of synapse activity,” says Professor Sauer.
Active zones are densely packed
The new knowledge was gained with the super-resolution microscopy method dSTORM (direct stochastic optical reconstruction microscopy). The method was developed by Sauer’s team in 2008. It enables individual molecules to be located even in the very small and densely packed active zones of synapses. This is not possible with conventional light microscopy because of the diffraction limit of 200 nanometers.
“For the first time we now have insights into the molecular organisation of the complex protein machines that control the signal transmission at the synapses of our brain,” says Professor Calebiro. Only with this knowledge will we be able to understand how the brain functions and how it processes information on different time scales.
The research teams will now use dSTORM to find out how all the proteins are distributed in the active synaptic zone. It is generally assumed that more than 100 proteins are involved in signal transmission in the active zones.
About this neuroscience research article
Source:
University of Wurzburg
Media Contacts:
Markus Sauer – University of Wurzburg
Image Source:
The image is credited to Chair Markus Sauer / University of Würzburg.
Original Research: Open access
“Super-resolution imaging reveals the nanoscale organization of metabotropic glutamate receptors at presynaptic active zones”. by Markus Sauer et al.
Science Advances doi:10.1126/sciadv.aay7193.
Abstract
Super-resolution imaging reveals the nanoscale organization of metabotropic glutamate receptors at presynaptic active zones
G protein–coupled receptors (GPCRs) play a fundamental role in the modulation of synaptic transmission. A pivotal example is provided by the metabotropic glutamate receptor type 4 (mGluR4), which inhibits glutamate release at presynaptic active zones (AZs). However, how GPCRs are organized within AZs to regulate neurotransmission remains largely unknown. Here, we applied two-color super-resolution imaging by direct stochastic optical reconstruction microscopy (dSTORM) to investigate the nanoscale organization of mGluR4 at parallel fiber AZs in the mouse cerebellum. We find an inhomogeneous distribution, with multiple nanodomains inside AZs, each containing, on average, one to two mGluR4 subunits. Within these nanodomains, mGluR4s are often localized in close proximity to voltage-dependent CaV2.1 channels and Munc-18-1, which are both essential for neurotransmitter release. These findings provide previously unknown insights into the molecular organization of GPCRs at AZs, suggesting a likely implication of a close association between mGluR4 and the secretory machinery in modulating synaptic transmission.
Feel Free To Share This Neuroscience News.