Summary: A new study sheds light on how we perceive and recognize specific visual stimuli.
Source: King’s College London.
Researchers from the Centre for Developmental Neurobiology at King’s College London have shed light on how we perceive and recognise specific visual stimuli.
The first steps in visual processing take place in the retina, which is a thin layer of tissue at the back of the eye containing more than 70 distinct types of nerve cells. Our visual system processes light stimuli to extract important information about the visual world, such as movement, orientation and colour. This information is processed at the cellular level before being transmitted via the optic nerve to the brain, where a visual image is formed.
Although some of the visual circuits underlying this image processing have been identified in previous research, this study is the first to reveal the cellular and molecular building blocks in the retina which enable us to detect oriented visual stimuli, a feature of visual perception described as ‘orientation selectivity’. Orientation selectivity is known to be important for various perceptual processes, including whether something is horizontal or vertical, which in humans is crucial for recognising faces.
Published today in Current Biology, the study shows how ‘orientation selectivity’ is orchestrated in the retina.
The researchers studied larval zebrafish, which has become an organism of choice for many scientists because its biological processes are relatively similar to humans. The development of zebrafish is also very fast, going from fertilised egg to larvae in five days – at which point they have a fully formed visual system, equivalent in overall structure and function to that of humans.
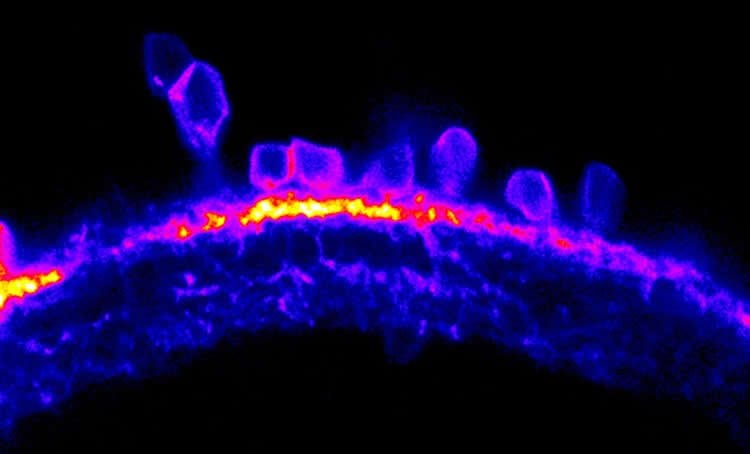
Crucially, larval zebrafish are also transparent, which means scientists can observe development and cellular processes as they happen, without using invasive experimental methods.
The zebrafish were shown videos featuring shapes of different orientation, while at the same time their brain activity was measured. The researchers analysed levels of cellular activity in neurons of the visual system by recording the fluorescence of calcium indicators (i.e. the more fluorescent the indicators were, the higher the level of neural activity). By doing so, it was possible to identify specific neurons in the retina and measure how they are activated by oriented stimuli.
Paride Antinucci, first author from the Centre for Developmental Neurobiology at King’s College London, said: ‘We heavily rely on our visual system to understand and interact with the world. A key property of visual perception is the ability of neural cells to selectively respond to oriented visual stimuli, a property called orientation selectivity. Our study reveals, for the first time, specific cell types and mechanisms responsible for the emergence of this property in the retina.’
Previous findings from the research team have shown a functional link between a protein called Teneurin-3 and orientation selectivity in the retina. This protein is thought to act as a recognition molecule at the communication junctions of neurons, called synapses. In this new study using novel genome editing technology, the researchers also discovered that mutating the Teneurin-3 gene led to a loss of orientation selectivity in neurons of the retina. More specifically, the retinal output neurons that send visual information to the brain, called ganglion cells, lost their ability to ‘see’ certain orientations when Teneurin-3 was not present.
The researchers also found that the structure of these special neurons played a crucial role in this process. The elongated shape of these cells directly determines if they are responding to oriented visual input and along which axis (i.e. vertical or horizontal). This ‘shape-determines-function’ mechanism is an important finding to explain visual processing.
Dr Robert Hindges, senior author from the Centre for Developmental Neurobiology at King’s, said: ‘For almost 20 years I’ve been studying topography – that is, how we see what is up and what is down, what is left and what is right. Today we are focusing on the investigation of visual processing in much more detail with the aid of zebrafish studies.
‘Our new research indicates that specifically shaped cells in the retina are activated when visual stimuli are horizontal, and other cells fire when they are vertical.’
Dr Hindges added: ‘These findings are important on two levels. Firstly, we finally get closer to understanding how different visual signals are shaped already in the retina before being transmitted to the brain. Secondly, our results help to explain how these types of proteins could work also in other contexts. Large genetic screens have previously shown that mutations in different members of the Teneurin gene family are strongly linked with mental health issues, bipolar disorder and epilepsy. We therefore believe our basic findings on the roles of these synaptic proteins could enhance our understanding of the development of these disorders.
Funding: This study was funded by the Biotechnology and Biological Sciences Research Council (BBSRC).
Source: Charlotte Hurley – King’s College London
Image Source: This NeuroscienceNews.com image is credited to King’s College London.
Original Research: Full open access research for “Neural Mechanisms Generating Orientation Selectivity in the Retina” by Paride Antinucci, Oniz Suleyman, Clinton Monfries, and Robert Hindges in Current Biology. Published online June 30 2016 doi:10.1016/j.cub.2016.05.035
[cbtabs][cbtab title=”MLA”]King’s College London. “Zebrafish Reveal the Ups and Downs of Vision.” NeuroscienceNews. NeuroscienceNews, 30 June 2016.
<https://neurosciencenews.com/vision-zebrafish-neuroscience-4609/>.[/cbtab][cbtab title=”APA”]King’s College London. (2016, June 30). Zebrafish Reveal the Ups and Downs of Vision. NeuroscienceNews. Retrieved June 30, 2016 from https://neurosciencenews.com/vision-zebrafish-neuroscience-4609/[/cbtab][cbtab title=”Chicago”]King’s College London. “Zebrafish Reveal the Ups and Downs of Vision.” https://neurosciencenews.com/vision-zebrafish-neuroscience-4609/ (accessed June 30, 2016).[/cbtab][/cbtabs]
Abstract
Neural Mechanisms Generating Orientation Selectivity in the Retina
Highlights
•We identify Tenm3+ ACs with elongated dendritic arbors showing orientation tuning
•Tenm3+ AC GABAergic inhibition is crucial for orientation-selective RGC tuning
•Orientation tuning is present also among some bipolar cell presynaptic terminals
•We propose a model of how orientation selectivity is generated in ganglion cells
Summary
The orientation of visual stimuli is a salient feature of visual scenes. In vertebrates, the first neural processing steps generating orientation selectivity take place in the retina. Here, we dissect an orientation-selective circuit in the larval zebrafish retina and describe its underlying synaptic, cellular, and molecular mechanisms. We genetically identify a class of amacrine cells (ACs) with elongated dendritic arbors that show orientation tuning. Both selective optogenetic ablation of ACs marked by the cell-adhesion molecule Teneurin-3 (Tenm3) and pharmacological interference with their function demonstrate that these cells are critical components for orientation selectivity in retinal ganglion cells (RGCs) by being a source of tuned GABAergic inhibition. Moreover, our morphological analyses reveal that Tenm3+ ACs and orientation-selective RGCs co-stratify their dendrites in the inner plexiform layer, and that Tenm3+ ACs require Tenm3 to acquire their correct dendritic stratification. Finally, we show that orientation tuning is present also among bipolar cell presynaptic terminals. Our results define a neural circuit underlying orientation selectivity in the vertebrate retina and characterize cellular and molecular requirements for its assembly.
“Neural Mechanisms Generating Orientation Selectivity in the Retina” by Paride Antinucci, Oniz Suleyman, Clinton Monfries, and Robert Hindges in Current Biology. Published online June 30 2016 doi:10.1016/j.cub.2016.05.035






