Summary: Stroking the skin can be as effective at relieving an itch as scratching, a new study reports. Rubbing an itch activates touch receptors and recruits anti-itch neurons in the spinal cord.
Source: SfN
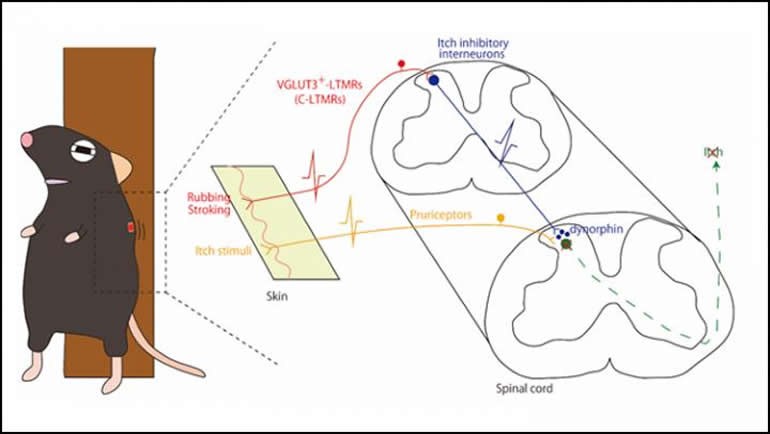
Stop scratching: rubbing skin activates an anti-itch pathway in the spinal cord, according to research in mice recently published in Journal of Neuroscience.
It can be hard to resist the relief of scratching an itch, even though scratching damages skin, especially in sensitive areas like the eyes. But stroking can relieve an itch, too. Sakai et al. investigated the neural pathway behind this less-damaging form of itch relief.
The research team triggered the urge to scratch in mice by administering an itch-inducing chemical underneath their skin. The team then recorded the electrical response from dorsal horn neurons in the spinal cord while they stroked the animals’ paws.
The neurons fired more often as the mice were stroked and less often after the stroking ended. These neurons respond to both touch and itch, so the increase corresponds to the added touch, not increased itchiness, while the decrease corresponds to itch relief.
The same decrease could be seen when the team directly stimulated touch-sensing neurons under the skin. However, inhibiting both sensory neurons and a subtype of anti-itch interneurons in the spinal cord failed to decrease the response from dorsal horn neurons, while activating sensory neurons stopped the mice from scratching.
The results show that stroking sets off a cascade, activating sensory neurons under the skin that then activate anti-itch interneurons in the spinal cord, resulting in reduced dorsal horn neural activity and itch relief.
About this neuroscience research article
Source:
SfN
Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Sakai et al., JNeurosci 2020.
Original Research: Closed access
“Low-Threshold Mechanosensitive VGLUT3-Lineage Sensory Neurons Mediate Spinal Inhibition of Itch by Touch” by Kent Sakai, Kristen M. Sanders, Shing-Hong Lin, Darya Pavlenko, Hideki Funahashi, Taisa Lozada, Shuanglin Hao, Chih-Cheng Chen and Tasuku Akiyama. Journal of Neuroscience.
Abstract
Low-Threshold Mechanosensitive VGLUT3-Lineage Sensory Neurons Mediate Spinal Inhibition of Itch by Touch
Innocuous mechanical stimuli, such as rubbing or stroking the skin, relieve itch through the activation of low-threshold mechanoreceptors (LTMRs). However, the mechanisms behind this inhibition remain unknown. We presently investigated whether stroking the skin reduces the responses of superficial dorsal horn neurons to pruritogens in male C57BL/6J mice. Single-unit recordings revealed that neuronal responses to chloroquine were enhanced during skin stroking, and this was followed by suppression of firing below baseline levels after the termination of stroking. Most of these neurons additionally responded to capsaicin. Stroking did not suppress neuronal responses to capsaicin, indicating state-dependent inhibition. Vesicular glutamate transporter 3 (VGLUT3)-lineage sensory nerves compose a subset of LTMRs. Stroking-related inhibition of neuronal responses to chloroquine was diminished by optogenetic inhibition of VGLUT3-lineage sensory nerves in male and female Vglut3-cre/NpHR-EYFP mice. Conversely, in male and female Vglut3-cre/ChR2-EYFP mice, optogenetic stimulation of VGLUT3-lineage sensory nerves inhibited firing responses of spinal neurons to pruritogens after the termination of stimulation. This inhibition was nearly abolished by spinal delivery of the κ-opioid receptor antagonist nor-binaltorphimine dihydrochloride, but not the neuropeptide Y receptor Y1 antagonist BMS193885. Optogenetic stimulation of VGLUT3-lineage sensory nerves inhibited pruritogen-evoked scratching without affecting mechanical and thermal pain behaviors. Therefore, VGLUT3-lineage sensory nerves appear to mediate inhibition of itch by tactile stimuli.
SIGNIFICANCE STATEMENT
Rubbing or stroking the skin is known to relieve itch. We investigated the mechanisms behind touch-evoked inhibition of itch in mice. Stroking the skin reduced the activity of itch-responsive spinal neurons. Optogenetic inhibition of VGLUT3-lineage sensory nerves diminished stroking-evoked inhibition, and optogenetic stimulation of VGLUT3-lineage nerves inhibited pruritogen-evoked firing. Together, our results provide a mechanistic understanding of touch-evoked inhibition of itch.






