Summary: Researchers have identified several novel functional mechanisms related to the inner hair cells of the ear. The findings may upend numerous longstanding theories about the working anatomical organization and workings of the ear, as well as provide new avenues for the development of technologies to improve hearing.
Source: Linköping University
Researchers at Linköping University, Sweden, have made several discoveries on the functioning mechanisms of the inner hair cells of the ear, which convert sounds into nerve signals that are processed in the brain.
The results, presented in the scientific journal Nature Communications, challenge the current picture of the anatomical organisation and workings of the hearing organ, which has prevailed for decades. A deeper understanding of how the hair cells are stimulated by sound is important for such matters as the optimisation of hearing aids and cochlear implants for people with hearing loss.
In order to hear sounds, we must convert sound waves, which are compressions and decompressions of air, into electrical nerve signals that are transmitted to the brain. This conversion takes place in the part of the inner ear known as the cochlea, due to its shape, which is reminiscent of a snail shell. The cochlear duct houses the hearing organ, with many hair cells that are divided into outer and inner hair cells.
The outer hair cells amplify sound vibrations, which enables us to hear faint sounds and perceive the various frequencies in human speech better. The inner hair cells convert the sound vibrations into nerve signals.
In the current study, the researchers have investigated how the conversion takes place. It is, namely, still unclear how the inner hair cells are stimulated by sound vibrations in order to produce nerve signals.
It has long been known that the outer hair cells are connected to a membrane that rests on top of them. The outer hair cells have hair-like protrusions known as stereocilia that are bent and activated when sound causes the membrane and the hearing organ to vibrate.
However, the current view is that the stereocilia of the inner hair cells are not in contact with this membrane, which is known as the tectorial membrane, and that they are stimulated by sounds by a completely different mechanism. It is this model that the new study challenges.
The relationship between the hair cells and the tectorial membrane has been studied in detail by electron microscopy since the 1950s. But it is extremely difficult to investigate how this gelatinous membrane functions, since it shrinks as soon as it is removed from the ear. This makes it extremely difficult to preserve the relationship between the inner hair cells and the tectorial membrane.
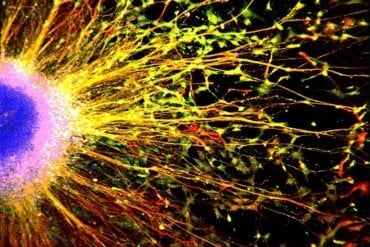
In addition, this membrane is transparent, and has therefore been essentially invisible. Until now. The LiU researchers noticed that the tectorial membrane reflected green light. This discovery made it possible to visualise the tectorial membrane by microscope.
“We cannot see any gap between the tectorial membrane and the hair cells. In contrast, the stereocilia on both outer and inner hair cells are completely embedded in the tectorial membrane. Our results are incompatible with the generally accepted idea that only the outer hair cells are in contact with the tectorial membrane”, says Pierre Hakizimana, principal research engineer at the Department of Biomedical and Clinical Sciences at Linköping University, and principal author of the article.
Pierre Hakizimana and his colleagues have studied the inner ear of guinea pigs, which is very similar to that of humans. When the researchers investigated the relationship between the membrane and the hair cells in more detail, they made a further discovery.
“We found calcium ducts with an appearance that we’ve never seen before. These calcium ducts span the tectorial membrane and connect to the stereocilia of both the inner and the outer hair cells”, says Pierre Hakizimana.
The research group, led by Professor Anders Fridberger, has previously discovered that the tectorial membrane functions as a reservoir for calcium ions, which are needed for the hair cells to convert the sound-evoked vibrations into nerve signals.
The researchers followed the motion of the calcium ions in the ducts, and their results suggest that the calcium ions flow through the ducts to the hair cells. This may explain how the hair cells obtain the large amounts of calcium ions needed for their function.
The study has also shown that the stereocilia on the inner and outer hair cells are bent by the tectorial membrane in similar ways. The next step of the research will be to understand in more detail how the calcium ions are transported, and identify the protein or proteins that make up the newly discovered calcium ducts.

“Our results allow us to describe a mechanism for how hearing functions, that is incompatible with the model that has been accepted for more than fifty years. The classic illustrations in the textbooks showing the hearing organ and how it functions must be updated. The mathematical models used in research to study hearing should also be updated to include these new findings”, says Pierre Hakizimana.
New information about how our hearing functions may in the long term be important for the development of cochlear implants. These are hearing aids that are inserted into the cochlea and which use electrical stimulation to make it possible for children and adults with hearing loss to perceive sounds.
“Cochlear implants are an amazing solution for treating hearing loss, but they can be improved. A deeper understanding of how the inner hair cells are stimulated by sounds is important to optimise how cochlear implants stimulate the auditory nerve”, says Pierre Hakizimana.
Funding: The study has received financial support from the Tysta Skolan Foundation, the Swedish Research Council, and the National Institutes of Health in the US.
About this auditory neuroscience research news
Source: Linköping University
Contact: Pierre Hakizimana – Linköping University
Image: The image is in the public domain
Original Research: Open access.
“Inner hair cell stereocilia are embedded in the tectorial membrane” by Pierre Hakizimana & Anders Fridberger. Nature Communications
Abstract
Inner hair cell stereocilia are embedded in the tectorial membrane
Mammalian hearing depends on sound-evoked displacements of the stereocilia of inner hair cells (IHCs), which cause the endogenous mechanoelectrical transducer channels to conduct inward currents of cations including Ca2+. Due to their presumed lack of contacts with the overlaying tectorial membrane (TM), the putative stimulation mechanism for these stereocilia is by means of the viscous drag of the surrounding endolymph.
However, despite numerous efforts to characterize the TM by electron microscopy and other techniques, the exact IHC stereocilia-TM relationship remains elusive.
Here we show that Ca2+-rich filamentous structures, that we call Ca2+ ducts, connect the TM to the IHC stereocilia to enable mechanical stimulation by the TM while also ensuring the stereocilia access to TM Ca2+.
Our results call for a reassessment of the stimulation mechanism for the IHC stereocilia and the TM role in hearing.