Summary: Researchers have found a way to produce realistic sensations of touch in two human amputees by directly stimulating the nervous system.
Source: University of Chicago.
Research provides blueprint for building neuroprosthetic devices that recreate the sense of touch by directly stimulating the nervous system.
Scientists at the University of Chicago and Case Western Reserve University have found a way to produce realistic sensations of touch in two human amputees by directly stimulating the nervous system.
The study, published Oct. 26 in Science Translational Medicine (STM), confirms earlier research on how the nervous system encodes the intensity, or magnitude, of sensations. It is the second of two groundbreaking publications this month by University of Chicago neuroscientist Sliman Bensmaia, PhD, using neuroprosthetic devices to recreate the sense of touch for amputee or quadriplegic patients with a “biomimetic” approach that approximates the natural, intact nervous system.
On Oct. 13, in a separate publication from STM, Bensmaia and a team led by Robert Gaunt, PhD, from the University of Pittsburgh, announced that for the first time, a paralyzed human patient was able to experience the sense of touch through a robotic arm that he controls with his brain. In that study, researchers interfaced directly with the patient’s brain, through an electrode array implanted in the areas of the brain responsible for hand movements and for touch, which allowed the man to both move the robotic arm and feel objects through it.
The new study takes a similar approach in amputees, working with two male subjects who each lost an arm after traumatic injuries. In this case, both subjects were implanted with neural interfaces, devices embedded with electrodes that were attached to the median, ulnar and radial nerves of the arm. Those are the same nerves that would carry signals from the hand were it still intact.
“If you want to create a dexterous hand for use in an amputee or a quadriplegic patient, you need to not only be able to move it, but have sensory feedback from it,” said Bensmaia, who is an associate professor of organismal biology and anatomy at the University of Chicago. “To do this, we first need to look at how the intact hand and the intact nervous system encodes this information, and then, to the extent that we can, try to mimic that in a neuroprosthesis.”
Recreating different sensations of intensity
The latest research is a joint effort by Bensmaia and Dustin Tyler, PhD, the Kent H. Smith Professor of Biomedical Engineering at Case Western Reserve University, who works with a large team trying to make bionic hands clinically viable. Tyler’s team, led by doctoral student Emily Graczyk, systematically tested the subjects’ ability to distinguish the magnitude of the sensations evoked when their nerves were stimulated through the interface. They varied aspects of the signals, such as frequency and intensity of each electrical pulse. The goal was to understand if there was a systematic way to manipulate the sensory magnitude.
Earlier research from Bensmaia’s lab predicted how the nervous system discerns intensity of touch, for example, how hard an object is pressing against the skin. That work suggested that the number of times certain nerve fibers fire in response to a given stimulus, known as the population spike rate, determines the perceived intensity of a given stimulus.
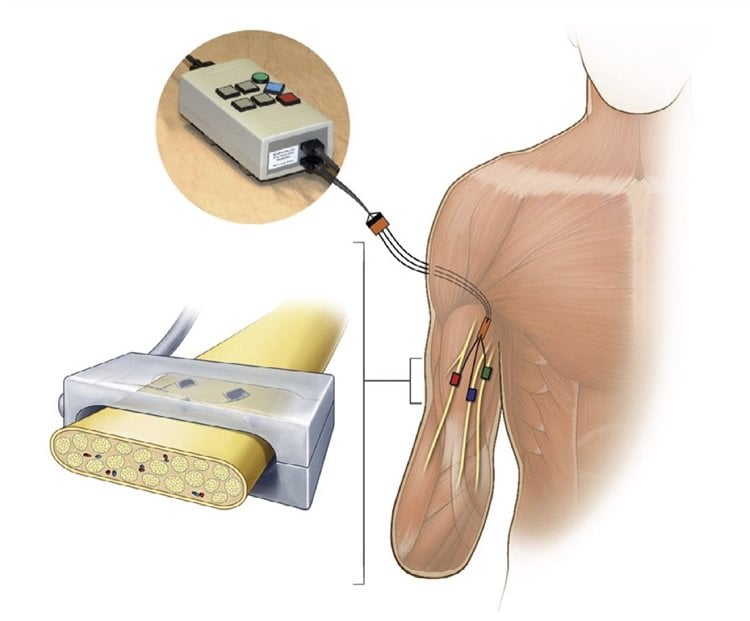
Results from the new study verify this hypothesis: A single feature of electrical stimulation—dubbed the activation charge rate—was found to determine the strength of the sensation. By changing the activation charge rate, the team could change sensory magnitude in a highly predictable way. The team then showed that the activation charge rate was also closely related to the evoked population spike rate.
Building neuroprosthetics that approximate the natural nervous system
While the new study furthers the development of neural interfaces for neuroprosthetics, artificial touch will only be as good as the devices providing input. In a separate paper published earlier this year in IEEE Transactions on Haptics, Bensmaia and his team tested the sensory abilities of a robotic fingertip equipped with touch sensors.
Using the same behavioral techniques that are used to test human sensory abilities, Bensmaia’s team, led by Benoit Delhaye and Erik Schluter, tested the finger’s ability to distinguish different touch locations, different pressure levels, the direction and speed of surfaces moving across it and the identity of textures scanned across it. The robotic finger (with the help of machine learning algorithms) proved to be almost as good as a human at most of these sensory tasks. By combining such high-quality input with the algorithms and data Bensmaia and Tyler produced in the other study, researchers can begin building neuroprosthetics that approximate natural sensations of touch.
Without realistic, natural-feeling sensations, neuroprosthetics will never come close to achieving the dexterity of our native hands. To illustrate the importance of touch, Bensmaia referred to a piano. Playing the piano requires a delicate touch, and an accomplished pianist knows how softly or forcefully to strike the keys based on sensory signals from the fingertips. Without these signals, the sounds the piano would make would not be very musical.
“The idea is that if we can reproduce those signals exactly, the amputee won’t have to think about it, he can just interact with objects naturally and automatically. Results from this study constitute a first step towards conveying finely graded information about contact pressure,” Bensmaia said.
Funding: The study, “The Neural Basis of Perceived Intensity in Natural and Artificial Touch,” was supported by the Defense Advanced Research Projects Agency (DARPA), the Department of Veterans Affairs, the National Science Foundation and the National Institutes of Health. Additional authors include Matthew Schiefer from the Louis-Stoke Veterans Affairs Medical Center, and Hannes Saal and Benoit Delhaye from the University of Chicago.
Source: Matt Wood – University of Chicago
Image Source: NeuroscienceNews.com image is credited to Graczyk et al, Sci. Transl. Med.
Original Research: Abstract for “Intracortical microstimulation of human somatosensory cortex” by Sharlene N. Flesher, Jennifer L. Collinger, Stephen T. Foldes, Jeffrey M. Weiss, John E. Downey, Elizabeth C. Tyler-Kabara, Sliman J. Bensmaia, Andrew B. Schwartz, Michael L. Boninger and Robert A. Gaunt in Science Translational Medicine. Published online October 19 2016 doi:10.1126/scitranslmed.aaf8083
[cbtabs][cbtab title=”MLA”]University of Chicago. “Restoring The Sense Of Touch In Amputees Using Natural Signals Of The Nervous System.” NeuroscienceNews. NeuroscienceNews, 26 October 2016.
<https://neurosciencenews.com/amputee-touch-sensation-5363/>.[/cbtab][cbtab title=”APA”]University of Chicago. (2016, October 26). Restoring The Sense Of Touch In Amputees Using Natural Signals Of The Nervous System. NeuroscienceNews. Retrieved October 26, 2016 from https://neurosciencenews.com/amputee-touch-sensation-5363/[/cbtab][cbtab title=”Chicago”]University of Chicago. “Restoring The Sense Of Touch In Amputees Using Natural Signals Of The Nervous System.” https://neurosciencenews.com/amputee-touch-sensation-5363/ (accessed October 26, 2016).[/cbtab][/cbtabs]
Abstract
Intracortical microstimulation of human somatosensory cortex
Intracortical microstimulation of the somatosensory cortex offers the potential for creating a sensory neuroprosthesis to restore tactile sensation. Whereas animal studies have suggested that both cutaneous and proprioceptive percepts can be evoked using this approach, the perceptual quality of the stimuli cannot be measured in these experiments. We show that microstimulation within the hand area of the somatosensory cortex of a person with long-term spinal cord injury evokes tactile sensations perceived as originating from locations on the hand and that cortical stimulation sites are organized according to expected somatotopic principles. Many of these percepts exhibit naturalistic characteristics (including feelings of pressure), can be evoked at low stimulation amplitudes, and remain stable for months. Further, modulating the stimulus amplitude grades the perceptual intensity of the stimuli, suggesting that intracortical microstimulation could be used to convey information about the contact location and pressure necessary to perform dexterous hand movements associated with object manipulation.
“Intracortical microstimulation of human somatosensory cortex” by Sharlene N. Flesher, Jennifer L. Collinger, Stephen T. Foldes, Jeffrey M. Weiss, John E. Downey, Elizabeth C. Tyler-Kabara, Sliman J. Bensmaia, Andrew B. Schwartz, Michael L. Boninger and Robert A. Gaunt in Science Translational Medicine. Published online October 19 2016 doi:10.1126/scitranslmed.aaf8083