Summary: In utero exposure to anesthesia appears to disrupt the migration of neurons to developing brain areas critical to language and cognition in rats.
Source: Rush University Medical Center
While there has long been discussion whether exposure to anesthesia affects brain development, new Rush University Medical Center research published in the journal Cerebral Cortex describes how prenatal anesthesia in rats seems to disrupt the “precisely choreographed” and timed migration of neurons to the area of the developing brain where thinking, memory and language occur.
The finding further fuels the debate about the safety of the anesthesia in pregnancy for unborn babies, with researchers suggesting the estimated 75,000 women undergo surgery while pregnant more closely examine surgery timing.
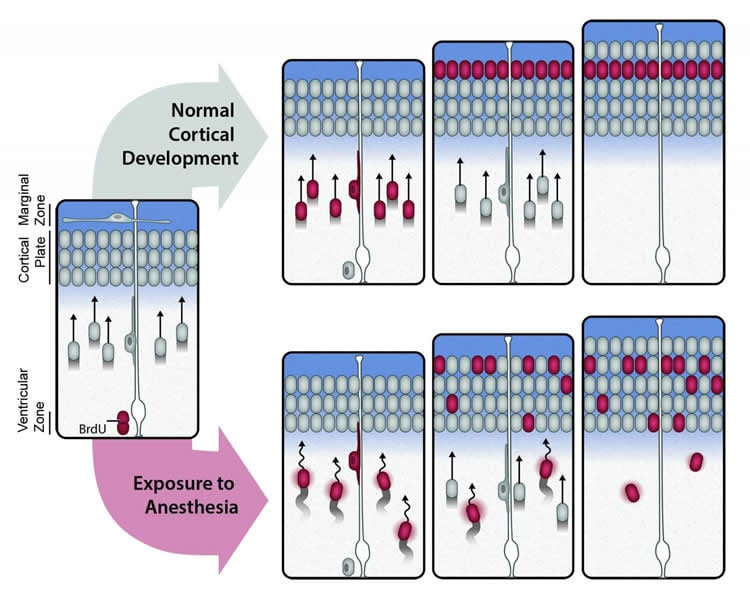
In a series of experiments detailed in the study In utero Exposure to Anesthetics Alters Neuronal Migration Pattern in Developing Cerebral Cortex and Causes Postnatal Behavioral Deficits in Rats, researchers demonstrated how exposing pregnant rats to anesthetics affects neuronal migration, the process in mammals in which neurons migrate from their original position deep inside the brain to their final position on the outer edge, or cerebral cortex.
“The cerebral cortex, or grey matter, is the brain’s computer processor. Cognitive processes like thinking, memory, and language are directed there, thus neurons never reaching their proper and predetermined positions in the cortex may have a profound impact on brain function” said the study’s lead author, anesthesiologist and neuroscientist Dr. Vicko Gluncic,MD, PhD. Dr. Gluncic conducted the research at Rush and now practices at Advocate Illinois Masonic Medical Center.
Anesthesia sends neurons down the wrong path
To test that hypothesis, researchers marked migrating neurons in rat fetuses with a dye and then anesthetized one group of pregnant rats during the precise period when neuronal migration occurs is its most active. That gestation period roughly corresponds to the late second trimester in humans – when motoric areas of the brain are already well developed while somatosensory and visual cortex is still developing. A control group was not anesthetized. Examinations of the rats exposed to anesthetics showed that significant number of neurons remained inappropriately scattered in deeper layers of cortex, and the longer the rats were exposed to prenatal anesthesia, the more scattered their neurons. A second set of behavioral experiments showed that all the littermates had consistent motor skills, but those exposed to anesthetics had significant behavioral and learnings deficits.
Together, the histological analysis of the rat brains and the behavioral testing demonstrates that in utero exposure to anesthesia negatively impacts formation of the most sensitive part of the brain by impeding neurons migration pattern. And in spite of these potent and potentially disastrous alterations, the authors say that the effects of anesthetics on neuronal migration and development of cortical columnar organization have never been tested before.
Mario Moric, a biostatistician in Rush’s Department of Anesthesiology, indicated that “there is a clear association of anesthesia type and anesthetic duration with neuronal migration and cognitive deficits at the drug levels used here (in rat fetuses), what is needed now is to further validate the mechanisms involved as well as test various anesthetics and anesthetic protocols to evaluate differential impact on cognitive functioning”
Researchers “don’t want to push the panic button” but push for more discussion
Study authors stressed that the finding in rats cannot translate directly to pregnant women cancelling or postponing needing surgery, though suggested the findings should be considered in inform discussions between women and their physicians.
“We don’t want to push the panic button,” Dr. Gluncic says. “It would be wrong if women delay or stop having medically indicated surgeries during pregnancy because of this. However, non-urgent surgeries in pregnancy are typically performed in the second trimester, after organogenesis, and when preterm contractions and spontaneous abortion are less likely than in last trimester. Based upon the present data, the possibility that anesthesia may affect fetal brain development should be seriously considered and disclosed in informed consent for anesthesia in the second trimester since the most active period of human fetal brain development actually occurs between 12th and 24th week of pregnancy.”
Source:
Rush University Medical Center
Media Contacts:
Charles Jolie – Rush University Medical Center
Image Source:
The image is credited to Dr. Vicko Gluncic, MD, PhD.
Original Research: Closed access.
“In utero Exposure to Anesthetics Alters Neuronal Migration Pattern in Developing Cerebral Cortex and Causes Postnatal Behavioral Deficits in Rats:
V Gluncic et al. Cerebral Cortex doi:10.1093/cercor/bhz065
Abstract
In utero Exposure to Anesthetics Alters Neuronal Migration Pattern in Developing Cerebral Cortex and Causes Postnatal Behavioral Deficits in Rats
During fetal development, cerebral cortical neurons are generated in the proliferative zone along the ventricles and then migrate to their final positions. To examine the impact of in utero exposure to anesthetics on neuronal migration, we injected pregnant rats with bromodeoxyuridine to label fetal neurons generated at embryonic Day (E) 17 and then randomized these rats to 9 different groups receiving 3 different means of anesthesia (oxygen/control, propofol, isoflurane) for 3 exposure durations (20, 50, 120 min). Histological analysis of brains from 54 pups revealed that significant number of neurons in anesthetized animals failed to acquire their correct cortical position and remained dispersed within inappropriate cortical layers and/or adjacent white matter. Behavioral testing of 86 littermates pointed to abnormalities that correspond to the aberrations in the brain areas that are specifically developing during the E17. In the second set of experiments, fetal brains exposed to isoflurane at E16 had diminished expression of the reelin and glutamic acid decarboxylase 67, proteins critical for neuronal migration. Together, these results call for cautious use of anesthetics during the neuronal migration period in pregnancy and more comprehensive investigation of neurodevelopmental consequences for the fetus and possible consequences later in life.






