Summary: Researchers have developed new software that is able to correctly map areas of the cortex.
Source: NIH/NIMH.
Software automatically detects each area’s ‘fingerprint’ in scans.
Researchers have mapped 180 distinct areas in our brain’s outer mantle, or cortex — more than twice the number previously known. They have also developed software that automatically detects the “fingerprint” of each of these areas in an individual’s brain scans. Funded by the National Institutes of Health through its Human Connectome Project (HCP), this software correctly mapped the areas by incorporating data from multiple non-invasive brain imaging measures that corroborated each other.
“These new insights and tools should help to explain how our cortex evolved and the roles of its specialized areas in health and disease, and could eventually hold promise for unprecedented precision in brain surgery and clinical work-ups,” said Bruce Cuthbert, Ph.D., acting director of NIH’s National Institute of Mental Health (NIMH), which co-funded the research as part of the HCP.
The new study identified — with a nearly 97 percent detection rate — 97 new cortex areas per hemisphere, in addition to confirming 83 that were previously known.
NIMH grantees David Van Essen, Ph.D., and Matthew Glasser, Ph.D., of Washington University in St. Louis, and colleagues at six other researcher centers, report on their discoveries July 20, 2016 in the journal Nature.
Earlier studies of cortex organization often used just one measure, such as examining postmortem tissue with a microscope. Uncertain delineation of cortex areas has sometimes led to shaky comparability of brain imaging findings.
“The situation is analogous to astronomy where ground-based telescopes produced relatively blurry images of the sky before the advent of adaptive optics and space telescopes,” noted Glasser, lead author of the study.
The HCP team set out to banish this blurriness by using multiple, precisely aligned, magnetic resonance imaging (MRI) modalities to measure cortical architecture, activity, connectivity, and topography in a group of 210 healthy participants. These measures — including cortex thickness, cortex myelin content, task and resting-state functional MRI (fMRI) – cross-validated each other. The findings were, in turn, confirmed in an additional independent sample of 210 healthy participants.
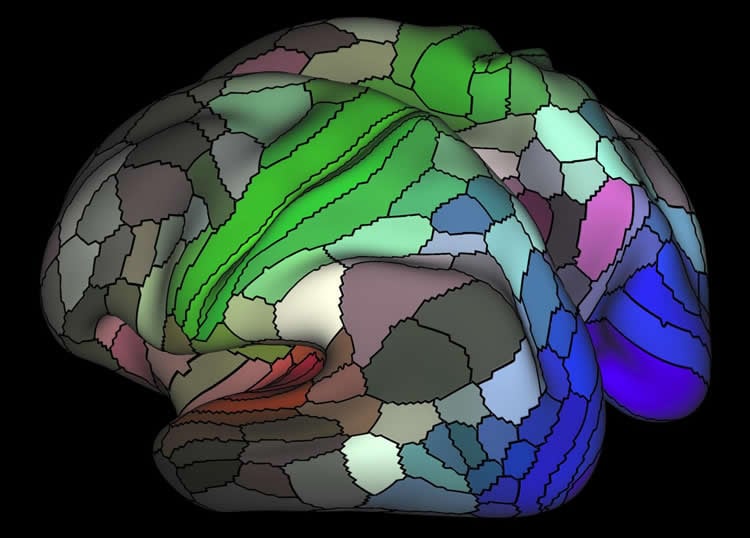
Even though some cortex areas turned out to be atypically located in a small minority of subjects, the data-derived algorithms incorporated into the software were able to successfully map them. While the study included fMRI scans of subjects performing tasks, the researchers determined that resting-state MRI techniques should suffice to map the areas in future studies using the tools they developed. Some areas may turn out to have further subdivisions or be subunits of other areas, in light of new data, noted senior author Van Essen.
“The ability to discriminate individual differences in the location, size, and topology of cortical areas from differences in their activity or connectivity should facilitate understanding of how each property is related to behavior and genetic underpinnings,” added Glasser.
The automated “areal classifier” and related tools are being shared with the research community via HCP websites. In addition, the extensively analyzed data underlying each of the published figures can be accessed via an NIH-funded database developed in the Van Essen laboratory.
Funding: The study was supported by National Institutes of Health, NIH/National Institute of Mental Health.
Source: Jules Asher – NIH/NIMH
Image Source: This NeuroscienceNews.com image is credited to Matthew Glasser, Ph.D., and David Van Essen, Ph.D., Washington University.
Original Research: Abstract for “A multi-modal parcellation of human cerebral cortex” by Matthew F. Glasser, Timothy S. Coalson, Emma C. Robinson, Carl D. Hacker, John Harwell, Essa Yacoub, Kamil Ugurbil, Jesper Andersson, Christian F. Beckmann, Mark Jenkinson, Stephen M. Smith and David C. Van Essen in Nature. Published online July 20 2016 doi:10.1038/nature18933
[cbtabs][cbtab title=”MLA”]NIH/NIMH. “Connectome Map More Than Doubles Human Cortex’s Known Regions.” NeuroscienceNews. NeuroscienceNews, 21 July 2016.
<https://neurosciencenews.com/connectome-map-cortex-4715/>.[/cbtab][cbtab title=”APA”]NIH/NIMH. (2016, July 21). Connectome Map More Than Doubles Human Cortex’s Known Regions. NeuroscienceNew. Retrieved July 21, 2016 from https://neurosciencenews.com/connectome-map-cortex-4715/[/cbtab][cbtab title=”Chicago”]NIH/NIMH. “Connectome Map More Than Doubles Human Cortex’s Known Regions.” https://neurosciencenews.com/connectome-map-cortex-4715/ (accessed July 21, 2016).[/cbtab][/cbtabs]
Abstract
A multi-modal parcellation of human cerebral cortex
Understanding the amazingly complex human cerebral cortex requires a map (or parcellation) of its major subdivisions, known as cortical areas. Making an accurate areal map has been a century-old objective in neuroscience. Using multi-modal magnetic resonance images from the Human Connectome Project (HCP) and an objective semi-automated neuroanatomical approach, we delineated 180 areas per hemisphere bounded by sharp changes in cortical architecture, function, connectivity, and/or topography in a precisely aligned group average of 210 healthy young adults. We characterized 97 new areas and 83 areas previously reported using post-mortem microscopy or other specialized study-specific approaches. To enable automated delineation and identification of these areas in new HCP subjects and in future studies, we trained a machine-learning classifier to recognize the multi-modal ‘fingerprint’ of each cortical area. This classifier detected the presence of 96.6% of the cortical areas in new subjects, replicated the group parcellation, and could correctly locate areas in individuals with atypical parcellations. The freely available parcellation and classifier will enable substantially improved neuroanatomical precision for studies of the structural and functional organization of human cerebral cortex and its variation across individuals and in development, aging, and disease.
“A multi-modal parcellation of human cerebral cortex” by Matthew F. Glasser, Timothy S. Coalson, Emma C. Robinson, Carl D. Hacker, John Harwell, Essa Yacoub, Kamil Ugurbil, Jesper Andersson, Christian F. Beckmann, Mark Jenkinson, Stephen M. Smith and David C. Van Essen in Nature. Published online July 20 2016 doi:10.1038/nature18933






