Patients suffering from hereditary neuropathy may have hope for new treatment thanks to a Geisinger study that uncovered a key to the development of the peripheral nervous system.
In an article published today in the online medical journal Nature Communications, Geisinger researchers found that a protein present within immune system cells plays a larger role than previously thought in the development of the peripheral nervous system.
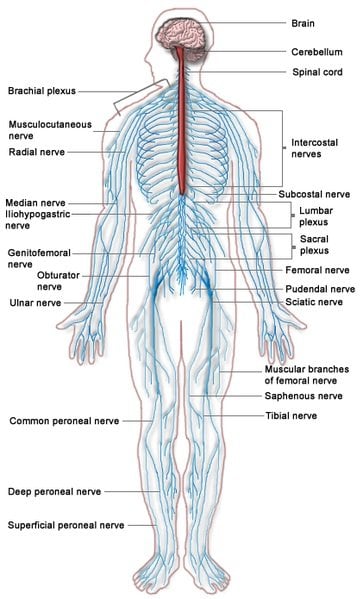
Nikolaos Tapinos, M.D., Ph.D., director of neurosurgery research and staff scientist at Geisinger’s Sigfried and Janet Weis Center for Research, said the findings could have implications in how hereditary neuropathy is treated. Hereditary neuropathy affects the peripheral nervous system, causing subtle symptoms such as muscle weakness, wasting and numbness that worsen over time.

“When the peripheral nervous system develops in utero, certain proteins control how the cells travel throughout the body to the proper locations,” Dr. Tapinos said. “Some of those proteins are already known, but this is the first time that the protein Lck has been identified as integral to this process.”
Lck, or lymphocyte-specific protein tyrosine kinase, is a protein that is found inside specialized cells of the immune system. Dr. Tapinos’ research found that Lck controls how cells called Schwann cells migrate across neurons throughout the peripheral nervous system.
Schwann cells function by creating the myelin sheath, the fatty covering that acts as an insulator around nerve fibers. In humans, the production of myelin begins in the 14th week of fetal development and continues through infancy and adolescence. When errors occur in the creation of myelin, hereditary neuropathy such as Charcot-Marie-Tooth disease (CMT), a motor and sensory neuropathy, can result.
“What we have found is that Lck is essentially the ‘switch’ that signals migration of the Schwann cells and production of the myelin sheath,” Dr. Tapinos said. “This finding sets the stage for further research into the specific molecular mechanisms that occur in order for this process to break down, and eventually toward developing treatments to prevent it.”
Notes about this neuropathy and neurology research
Other authors on the article are Jennifer Ness Myers and Kristin Snyder, both of Geisinger’s Sigfried and Janet Weis Center for Research.
Contact: Amanda O’Rourke – Geisinger Health System
Source: Geisinger Health System press release
Image Source: The nervous system diagram is available in the public domain.
Original Research: Full open access research for “Lck tyrosine kinase mediates β1-integrin signalling to regulate Schwann cell migration and myelination” by Jennifer K. Ness, Kristin M. Snyder and Nikos Tapinos in Nature Communications. Published online May 28 2013 doi:10.1038/ncomms2928