Summary: EGR3 and BDNF may play a critical role in the impaired cellular resilience seen in bipolar disorder, researchers report.
Source: PCC.
Bipolar Disorder (BD) is a multifactorial brain disorder in which patients experience radical shifts in mood and undergo periods of depression followed by periods of mania. It has been known for some time that both environmental and genetic factors play important roles in the disease. For instance, being exposed to high levels of stress for long periods, and especially during childhood, has been associated with the development of BD.
Immediate early genes (IEGs) are a class of genes that respond very rapidly to environmental stimuli, and that includes stress. IEGs respond to a stressor by activating other genes that lead to neuronal plasticity, the ability of brain cells to change in form and function in response to changes in the environment. Ultimately, it is the process of neuronal plasticity that gives the brain the ability to learn from and adapt to new experiences.
One type of protein produced by IEGs is the so-called Early Growth Response (EGR) proteins, which translate environmental influence into long-term changes in the brain. These proteins are found throughout the brain and are highly produced in response to environmental changes such as stressful stimuli and sleep deprivation. Without the action played out by these proteins, brain cells and the brain itself cannot appropriately respond to the many stimuli that are constantly received from the environment.
Effective neuronal plasticity also depends on neurotrophins, which are regulatory factors that promote development and survival of brain cells. Brain-derived neurotrophic factor (BDNF) is the neurotrophin mostly found in the brain. It has been extensively investigated in BD patients and has been suggested as a hallmark of BD. Indeed, some studies have shown that the levels of BDNF in the serum of BD patients are reduced whenever patients undergo a period of depression, hypomania, or mania. Other studies have shown that regardless of mood state, BD patients present reduced levels of BDNF. Overall, changes in BDNF levels seem to be a characteristic found in BD patients that may contribute to the pathophysiology of the disease.
Now an international team of researchers from Universidade Federal do Rio Grande do Sul in Brazil, University of Arizona College of Medicine in the United States and McMaster University in Canada have published an article connecting the dots between these two players to explain the impaired cellular resilience observed in BD that in the grand scheme of things may relate to the impaired resilience presented by BD patients to respond to events, including stress.
In a previous study done by the group in 2016, one type of IEG gene known as EGR3, that normally responds to environmental events and stressful stimuli, was found repressed in the brain of BD patients, suggesting that when facing a stressor, the EGR3 in BD patients does not respond to the stimulus appropriately. Indeed, BD patients are highly prone to stress and have more difficulties dealing with stress or adapting to it if compared to healthy individuals. What the research group is now suggesting is that both EGR3 and BDNF may each play a critical role in the impaired cellular resilience seen in BD, and that each of these two genes may affect each other’s expression in the cell. “We believe that the reduced level of BDNF that has been extensively observed in BD patients is caused by the fact that EGR3 is repressed in the brain of BD patients. The two molecules are interconnected in a regulatory pathway that is disrupted in BD patients,” says Fabio Klamt, leading author of the article entitled “EGR3 immediate early gene and the brain-derived neurotrophic factor in bipolar disorder” and published on February 5th in the journal Frontiers in Behavioral Neuroscience.
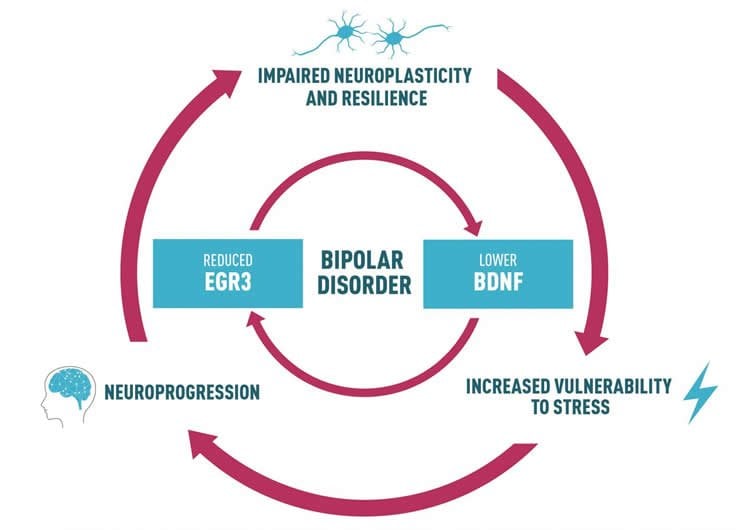
The authors also add that the fact that EGR3 responds very quickly to environmental stimuli renders the molecule a potential drug target. “It is possible to imagine that EGR3 may be modulated in order to increase its expression and that of BDNF, which may have a positive impact on BD patients,” says Bianca Pfaffenseller, a scientist working at Hospital de Clínicas de Porto Alegre, in Brazil, and the first author of the study.
The idea that mental disorders should be seen as any other chronic disease in which the underlying biology plays an important role has replaced the old descriptions of mental illnesses as the result of bad psychological influences. As Nobel prize laureate Eric Kandel has said, “all mental processes are brain processes and therefore all disorders of mental functioning are biological diseases.” The perspective article authored by Fabio Klamt and colleagues supports this view by offering new insights into the underlying biology of this lifelong and devastating mental disorder affecting millions of people worldwide.
Funding: The study was supported by the Brazilian grant CNPq/MS/SCTIE/DECIT – Pesquisas Sobre Doenças Neurodegenerativas (466989/2014-8) and INCT-TM/CNPq/FAPESP (#465458/2014-9).
Source: Fabio Klamt – PCC
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Cezar Farias.
Original Research: Open access research in Frontiers in Behavioral Neuroscience.
doi:10.3389/fnbeh.2018.00015
[cbtabs][cbtab title=”MLA”]PCC “New Path For Bipolar Disorder Comes to Light.” NeuroscienceNews. NeuroscienceNews, 5 February 2018.
<https://neurosciencenews.com/biology-bipolar-disorder-8429/>.[/cbtab][cbtab title=”APA”]PCC (2018, February 5). New Path For Bipolar Disorder Comes to Light. NeuroscienceNews. Retrieved February 5, 2018 from https://neurosciencenews.com/biology-bipolar-disorder-8429/[/cbtab][cbtab title=”Chicago”]PCC “New Path For Bipolar Disorder Comes to Light.” https://neurosciencenews.com/biology-bipolar-disorder-8429/ (accessed February 5, 2018).[/cbtab][/cbtabs]
Abstract
EGR3 Immediate Early Gene and the Brain-Derived Neurotrophic Factor in Bipolar Disorder
Bipolar disorder (BD) is a severe psychiatric illness with a consistent genetic influence, involving complex interactions between numerous genes and environmental factors. Immediate early genes (IEGs) are activated in the brain in response to environmental stimuli, such as stress. The potential to translate environmental stimuli into long-term changes in brain has led to increased interest in a potential role for these genes influencing risk for psychiatric disorders. Our recent finding using network-based approach has shown that the regulatory unit of early growth response gene 3 (EGR3) of IEGs family was robustly repressed in postmortem prefrontal cortex of BD patients. As a central transcription factor, EGR3 regulates an array of target genes that mediate critical neurobiological processes such as synaptic plasticity, memory and cognition. Considering that EGR3 expression is induced by brain-derived neurotrophic factor (BDNF) that has been consistently related to BD pathophysiology, we suggest a link between BDNF and EGR3 and their potential role in BD. A growing body of data from our group and others has shown that peripheral BDNF levels are reduced during mood episodes and also with illness progression. In this same vein, BDNF has been proposed as an important growth factor in the impaired cellular resilience related to BD. Taken together with the fact that EGR3 regulates the expression of the neurotrophin receptor p75NTR and may also indirectly induce BDNF expression, here we propose a feed-forward gene regulatory network involving EGR3 and BDNF and its potential role in BD.






