Summary: Study provides potential new avenues to treat Huntington’s disease and other neurodegenerative disorders.
Source: SBP.
Research identifies cellular recycling process linked to beneficial effects of enduring mild stress.
Biologists have known for decades that enduring a short period of mild stress makes simple organisms and human cells better able to survive additional stress later in life. Now, scientists at Sanford Burnham Prebys Medical Discovery Institute (SBP) have found that a cellular process called autophagy is critically involved in providing the benefits of temporary stress. The study, published today in Nature Communications, creates new avenues to pursue treatments for neurological disorders such as Huntington’s disease.
Autophagy is a means of recycling cells’ old, broken, or unneeded parts so that their components can be re-used to make new molecules or be burned for energy. The process had previously been linked to longevity, in no small part because of research led by Malene Hansen, Ph.D., associate professor at SBP and senior author of the study. The new results suggest that long life and stress resistance are connected at the cellular level.
“We used C. elegans–tiny roundworms used to study fundamental biology–to test the importance of autophagy in becoming stress resistant,” says Caroline Kumsta, Ph.D., staff scientist in Hansen’s lab and lead author of the study. “They’re a great model system because they’re transparent, so you can easily observe what goes on inside them, most of their genes and molecular signaling pathways have functional counterparts in humans, and they only live a few weeks, which greatly facilitate measuring their lifespans.”
Kumsta and colleagues incubated worms at 36 °C, significantly above the temperature they are usually kept at in the laboratory, for one hour. After this short heat exposure–a mild form of stress that improves the organism’s survival–autophagy rates increased throughout the worms’ tissues. When they exposed these heat-primed worms to another, longer heat shock a few days later, worms that were deficient in autophagy failed to benefit from the initial mild heat shock, as observed in heat-primed worms with intact autophagy.
The researchers reasoned that a mild heat stress might also improve the worms’ ability to handle another condition that worsens with age–buildup of aggregated proteins, which is stressful for cells. To test this hypothesis, Kumsta used worms that model Huntington’s disease, a fatal inherited disorder caused by neuronal proteins that start to stick together into big clumps as patients age, leading to degeneration throughout the brain. Exposing worms that make similar sticky proteins in different tissues to a mild heat shock reduced the number of protein aggregates, suggesting that a limited amount of heat stress can reduce toxic protein aggregation.
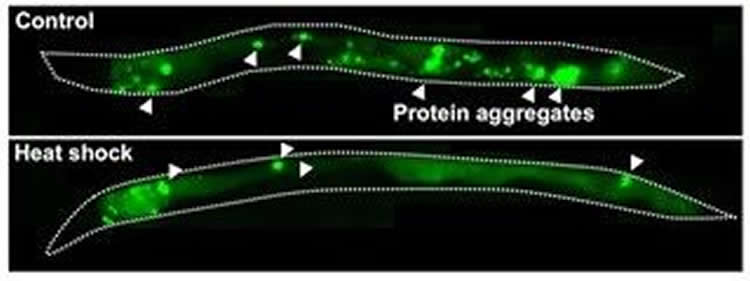
“Our finding that brief heat exposure helps alleviate protein aggregation is exciting because it could lead to new approaches to slow the advance of neurodegenerative diseases such as Huntington’s,” says Hansen. “The results may also be relevant to Alzheimer’s and Parkinson’s, which are similarly caused by clumping-prone proteins.”
“This research raises many exciting questions,” adds Hansen. “For example, how does induction of autophagy by a mild heat stress early on make cells better able to survive heat later–what’s the cellular memory? There’s a lot to follow up on.”
“A lot of people ask us if this means they should start going to the sauna or do hot yoga,” jokes Kumsta. “That may not be an entirely bad idea–epidemiological studies do indicate that frequent sauna use is associated with longer life. But we have a lot more research to do to figure out whether that has anything to do with the beneficial induction of autophagy by heat stress that we see in C. elegans.”
Kumsta recently received a promotion from postdoc to staff scientist in recognition of her leadership of this study.
Funding: Financial support for the study was provided by the American Federation for Aging Research and the National Institutes of Health.
Source: Susan Gammon – SBP
Image Source: NeuroscienceNews.com image is credited to Caroline Kumsta, Ph.D.
Original Research: Full open access research for “Hormetic heat stress and HSF-1 induce autophagy to improve survival and proteostasis in C. elegans” by Caroline Kumsta, Jessica T. Chang, Jessica Schmalz & Malene Hansen Nature Communications. Published online February 15 2017 doi:10.1038/ncomms14337
[cbtabs][cbtab title=”MLA”]SBP “What Doesn’t Kill You Makes You Stronger.” NeuroscienceNews. NeuroscienceNews, 15 February 2017.
<https://neurosciencenews.com/autophagy-stress-neurology-6115/>.[/cbtab][cbtab title=”APA”]SBP (2017, February 15). What Doesn’t Kill You Makes You Stronger. NeuroscienceNew. Retrieved February 15, 2017 from https://neurosciencenews.com/autophagy-stress-neurology-6115/[/cbtab][cbtab title=”Chicago”]SBP “What Doesn’t Kill You Makes You Stronger.” https://neurosciencenews.com/autophagy-stress-neurology-6115/ (accessed February 15, 2017).[/cbtab][/cbtabs]
Abstract
Hormetic heat stress and HSF-1 induce autophagy to improve survival and proteostasis in C. elegans
Stress-response pathways have evolved to maintain cellular homeostasis and to ensure the survival of organisms under changing environmental conditions. Whereas severe stress is detrimental, mild stress can be beneficial for health and survival, known as hormesis. Although the universally conserved heat-shock response regulated by transcription factor HSF-1 has been implicated as an effector mechanism, the role and possible interplay with other cellular processes, such as autophagy, remains poorly understood. Here we show that autophagy is induced in multiple tissues of Caenorhabditis elegans following hormetic heat stress or HSF-1 overexpression. Autophagy-related genes are required for the thermoresistance and longevity of animals exposed to hormetic heat shock or HSF-1 overexpression. Hormetic heat shock also reduces the progressive accumulation of PolyQ aggregates in an autophagy-dependent manner. These findings demonstrate that autophagy contributes to stress resistance and hormesis, and reveal a requirement for autophagy in HSF-1-regulated functions in the heat-shock response, proteostasis and ageing.
“Hormetic heat stress and HSF-1 induce autophagy to improve survival and proteostasis in C. elegans” by Caroline Kumsta, Jessica T. Chang, Jessica Schmalz & Malene Hansen Nature Communications. Published online February 15 2017 doi:10.1038/ncomms14337